Figure 1
Download original image
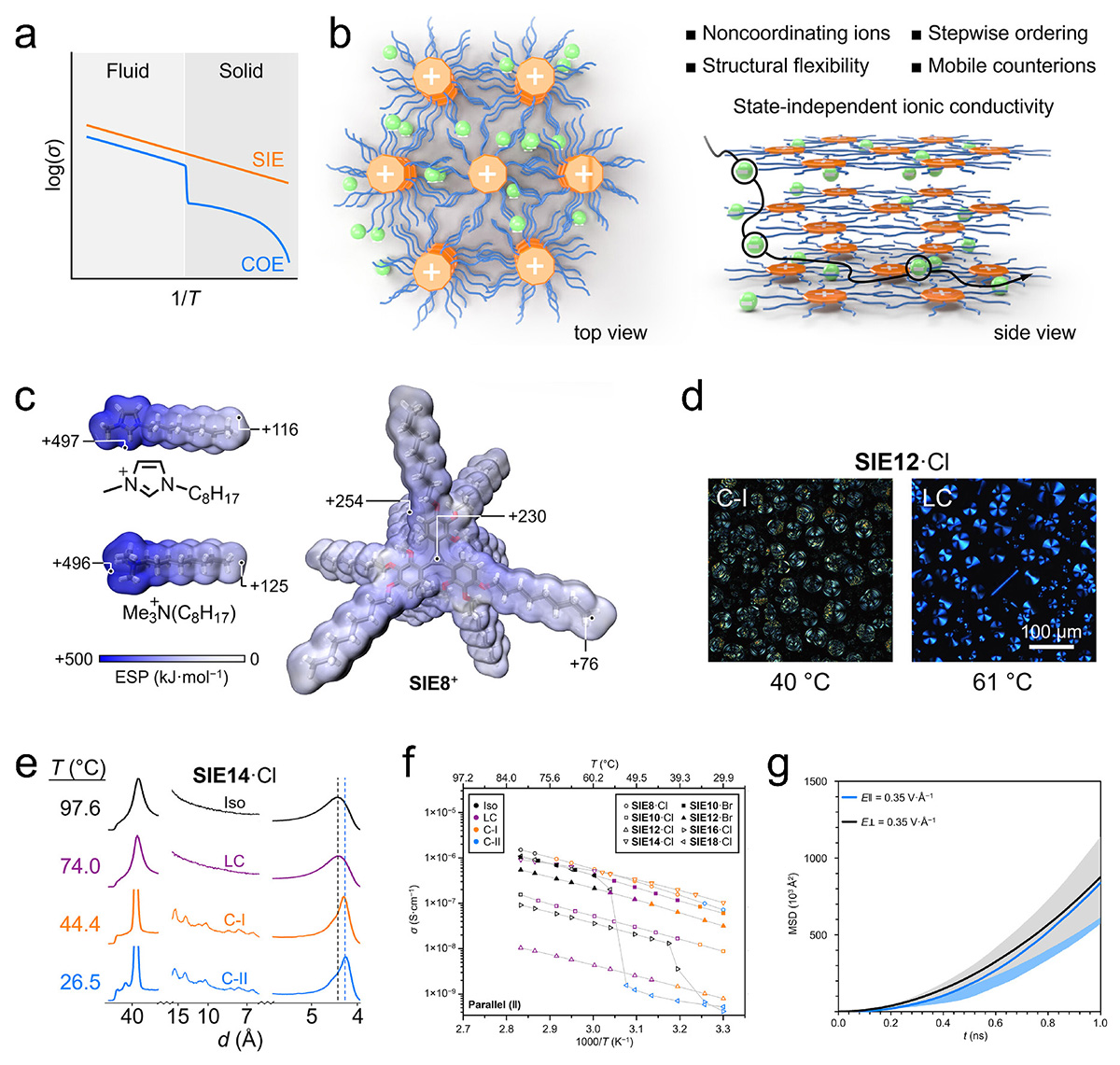
Structural design, phase transition behavior, and ionic transport properties of state-independent electrolytes. (a) COEs show an abrupt conductivity drop at the fluid-solid transition, whereas SIEs maintain continuous Arrhenius behavior across phases. (b) SIE superstructure: mobile ions diffuse isotropically through an anisotropic columnar network of noncoordinating cations; positional and orientational order develops stepwise while alkyl chains remain flexible. (c) Electrostatic potential maps of SIEn+ versus imidazolium and ammonium cations, showing extensive charge delocalization and threefold symmetry. (d) POM images of SIE12·Cl as a crystal (C-I) at 40 °C and showing a characteristic Colhex liquid crystal texture at 61 °C. (e) Variable-temperature X-ray scattering patterns of SIE14·Cl, revealing differences in long- and short-range order across its Iso, LC, C-I, and C-II phases. The intensity in the d-range from ~6 to 15 Å is scaled up to visualize low-intensity peaks, with dashed lines overlaid to facilitate spectral comparison. (f) DC conductivity from the Iso to LC or C-I phases (n = 8, 10, 12, and 14 carbon atoms) shows continuous Arrhenius behavior; from Iso to the C-II phase (n = 16 and 18 carbon atoms) show a sharp drop. (g) MD simulations of Cl− MSD in SIE10·Cl crystal under parallel and perpendicular fields, demonstrating isotropic ion transport. Adapted with permission from Ref. [7].
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.

