| Issue |
Natl Sci Open
Volume 5, Number 2, 2026
|
|
|---|---|---|
| Article Number | 20260009 | |
| Number of page(s) | 3 | |
| Section | Materials Science | |
| DOI | https://doi.org/10.1360/nso/20260009 | |
| Published online | 19 January 2026 | |
COMMENTARY
Continuous ionic conductivity across phase boundaries in organic electrolytes
College of Materials Science and Engineering, Hunan Joint International Laboratory of Advanced Materials and Technology for Clean Energy, Hunan Province Key Laboratory for Advanced Carbon Materials and Applied Technology, Hunan University, Changsha 410082, China
* Corresponding author (email: This email address is being protected from spambots. You need JavaScript enabled to view it.
)
Received:
14
January
2026
Accepted:
15
January
2026
The sharp decline in ionic conductivity at the liquid-solid phase transition is an intrinsic limitation of organic electrolytes. In conventional organic electrolytes (COEs), crystallization triggers pronounced conductivity loss and deviates from Arrhenius behavior, reflecting a fundamental shift in ion-transport mechanisms (Figure 1a) [1,2]. In contrast, inorganic superionic conductors sustain fast ion transport in crystalline lattices when migration barriers are low and diffusion sites are energetically comparable [3,4]. Replicating this in organic systems remains challenging, as crystallization strengthens ion pairing and restricts molecular mobility [5], while structural heterogeneity disrupts transport continuity—a universal electrolyte design challenge also observed in lithium-ion battery electrolytes with solvation structure imbalance under cryogenic conditions [6].
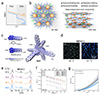 |
Figure 1 Structural design, phase transition behavior, and ionic transport properties of state-independent electrolytes. (a) COEs show an abrupt conductivity drop at the fluid-solid transition, whereas SIEs maintain continuous Arrhenius behavior across phases. (b) SIE superstructure: mobile ions diffuse isotropically through an anisotropic columnar network of noncoordinating cations; positional and orientational order develops stepwise while alkyl chains remain flexible. (c) Electrostatic potential maps of SIEn+ versus imidazolium and ammonium cations, showing extensive charge delocalization and threefold symmetry. (d) POM images of SIE12·Cl as a crystal (C-I) at 40 °C and showing a characteristic Colhex liquid crystal texture at 61 °C. (e) Variable-temperature X-ray scattering patterns of SIE14·Cl, revealing differences in long- and short-range order across its Iso, LC, C-I, and C-II phases. The intensity in the d-range from ~6 to 15 Å is scaled up to visualize low-intensity peaks, with dashed lines overlaid to facilitate spectral comparison. (f) DC conductivity from the Iso to LC or C-I phases (n = 8, 10, 12, and 14 carbon atoms) shows continuous Arrhenius behavior; from Iso to the C-II phase (n = 16 and 18 carbon atoms) show a sharp drop. (g) MD simulations of Cl− MSD in SIE10·Cl crystal under parallel and perpendicular fields, demonstrating isotropic ion transport. Adapted with permission from Ref. [7]. |
In a recent Science study, Barclay et al. [7] introduced a new class of state-independent electrolytes (SIEs) that challenge this conventional view. These SIEs maintain the same ion conduction mechanism across isotropic liquid, liquid-crystalline, and crystalline solid phases. As illustrated in Figure 1a, unlike COEs, they avoid the abrupt loss in conductivity and instead show smooth Arrhenius behavior across phase transitions.
This unusual behavior stems from a triskelion-like tris(trialkoxyphenyl) cyclopropenium structure (Figure 1b), which integrates three essential design elements: weak cation-anion interactions, a solid-state framework offering multiple energetically near-equivalent sites for counterions, and retained structural flexibility across phase transitions. Electrostatic potential maps (Figure 1c) show extensive charge delocalization over the cyclopropenium core, significantly weakening ion-pair interactions compared with conventional ammonium and imidazolium cations, providing a molecular basis for the state-independent ion transport observed in SIEs.
Consistent with this molecular design, SIEs exhibit stepwise structural ordering upon cooling. Polarized optical microscopy (Figure 1d) reveals an intermediate hexagonal columnar liquid-crystalline phase between the isotropic liquid and crystalline solid states, allowing the lattice to form gradually rather than abruptly. Variable-temperature X-ray scattering of SIE14·Cl (Figure 1e) provides further evidence of stepwise structural ordering. The diffraction peaks gradually sharpen and new peaks emerge as the material evolves from the isotropic liquid (Iso) via the liquid-crystalline (LC) phase to the crystalline C-I and C-II phases, and a persistent shoulder at ~4.5 Å indicates that alkyl chains retain substantial flexibility even in the solid state, which is consistent with the preserved molecular dynamics across all phases.
As a result, ionic conductivity follows a continuous Arrhenius trend across all phases, with nearly constant activation energies for different SIE compositions (Figure 1f). Notably, despite the anisotropic columnar packing of the cations, ion transport remains isotropic. Molecular dynamics simulations further reveal that halide ions exhibit comparable diffusion parallel and perpendicular to the columnar axis (Figure 1g), demonstrating that ionic motion is not constrained by the underlying structural anisotropy.
In summary, Barclay et al. present a molecular-level design principle that breaks the long-standing constraint between crystallization and ionic transport in organic electrolytes. By maintaining the same conduction mechanism through all phase transitions, this work establishes a general “charge delocalization-stepwise ordering-transport continuity” framework, providing a new conceptual foundation for designing solid-state and phase-tolerant organic electrolytes.
Conflict of interest
The authors declare no conflict of interest.
References
- MacFarlane DR, Huang J, Forsyth M. Lithium-doped plastic crystal electrolytes exhibiting fast ion conduction for secondary batteries. Nature 1999; 402: 792-794. [Article] [Google Scholar]
- Zhao Q, Stalin S, Zhao CZ, et al. Designing solid-state electrolytes for safe, energy-dense batteries. Nat Rev Mater 2020; 5: 229-252. [Article] [NASA ADS] [CrossRef] [Google Scholar]
- Zeng Y, Ouyang B, Liu J, et al. High-entropy mechanism to boost ionic conductivity. Science 2022; 378: 1320-1324. [Article] [Google Scholar]
- Han G, Vasylenko A, Daniels LM, et al. Superionic lithium transport via multiple coordination environments defined by two-anion packing. Science 2024; 383: 739-745. [Article] [Google Scholar]
- Chen X, Yao N, Zheng Z, et al. A perspective on the fundamental theory of nonaqueous electrolytes for rechargeable batteries. Natl Sci Rev 2025; 12: nwae394. [Article] [Google Scholar]
- Chen Y, Wang A, Zhao Y, et al. Decoding polarity gradient enabled ultra-high lithium ion conduction. Natl Sci Rev 2026; 13: nwaf543. [Article] [Google Scholar]
- Barclay J, Williamson JM, Litt H, et al. State-independent ionic conductivity. Science 2025; 390: 1254-1258. [Article] [Google Scholar]
© The Author(s) 2026. Published by Science Press and EDP Sciences.
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
All Figures
 |
Figure 1 Structural design, phase transition behavior, and ionic transport properties of state-independent electrolytes. (a) COEs show an abrupt conductivity drop at the fluid-solid transition, whereas SIEs maintain continuous Arrhenius behavior across phases. (b) SIE superstructure: mobile ions diffuse isotropically through an anisotropic columnar network of noncoordinating cations; positional and orientational order develops stepwise while alkyl chains remain flexible. (c) Electrostatic potential maps of SIEn+ versus imidazolium and ammonium cations, showing extensive charge delocalization and threefold symmetry. (d) POM images of SIE12·Cl as a crystal (C-I) at 40 °C and showing a characteristic Colhex liquid crystal texture at 61 °C. (e) Variable-temperature X-ray scattering patterns of SIE14·Cl, revealing differences in long- and short-range order across its Iso, LC, C-I, and C-II phases. The intensity in the d-range from ~6 to 15 Å is scaled up to visualize low-intensity peaks, with dashed lines overlaid to facilitate spectral comparison. (f) DC conductivity from the Iso to LC or C-I phases (n = 8, 10, 12, and 14 carbon atoms) shows continuous Arrhenius behavior; from Iso to the C-II phase (n = 16 and 18 carbon atoms) show a sharp drop. (g) MD simulations of Cl− MSD in SIE10·Cl crystal under parallel and perpendicular fields, demonstrating isotropic ion transport. Adapted with permission from Ref. [7]. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.

