| Issue |
Natl Sci Open
Volume 5, Number 2, 2026
|
|
|---|---|---|
| Article Number | 20250043 | |
| Number of page(s) | 25 | |
| Section | Earth and Environmental Sciences | |
| DOI | https://doi.org/10.1360/nso/20250043 | |
| Published online | 27 November 2025 | |
REVIEW
Piezoelectrotrophy: A new paradigm for microbial energy acquisition
1
Fujian Provincial Key Laboratory of Soil Environmental Health and Regulation, College of Resources and Environment, Fujian Agriculture and Forestry University, Fuzhou 350002, China
2
College of Biomedicine, Fujian Agriculture and Forestry University, Fuzhou 350002, China
* Corresponding author (email: This email address is being protected from spambots. You need JavaScript enabled to view it.
)
Received:
3
September
2025
Revised:
13
November
2025
Accepted:
25
November
2025
Abstract
This review introduces piezoelectrotrophy as a novel paradigm in microbial energy acquisition, complementing phototrophy, chemotrophy, and electrotrophy. We define piezoelectrotrophy as the process enabling microorganisms to harness mechanical energy via piezoelectric mechanisms, converting it into electrical energy for cellular metabolism. This expands microbial energy utilization beyond light and chemical sources to ubiquitous mechanical energy in natural systems. We propose a framework for piezoelectromicrobiology, emphasizing the nano-bio interface for electron generation, transfer, and uptake. The concept carries profound ecological and evolutionary implications, from cellular metabolism to ecosystem dynamics. We explore applications in environmental remediation, sensing technologies, bioelectronics, and medical implants. A roadmap addresses methodological challenges and suggests validation experiments. Piezoelectrotrophy offers a conceptual framework for investigating mechanical energy use in microbial systems, providing testable hypotheses for energy acquisition in dark, oligotrophic environments, biogeochemical cycles, microbial evolution, and biotechnology. Rigorous experimental validation is essential to confirm its ecological relevance and energetic viability.
Key words: piezoelectrotrophy / microbial energetics / piezoelectricity / mechanical energy / extracellular electron transfer / bioelectrochemistry
© The Author(s) 2025. Published by Science Press and EDP Sciences.
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
INTRODUCTION
The fundamental question of how life sustains itself has driven microbiological research for centuries, revealing sophisticated mechanisms by which microorganisms extract energy from their environment. Current understanding encompasses three primary energy-harvesting paradigms that have shaped microbial ecology and metabolism (Figure 1A–C). These established frameworks, while comprehensive in their respective domains, may not capture the full spectrum of energy utilization strategies available to microbial life.
 |
Figure 1 Types of energy utilization by microorganisms. (A) Phototrophy; (B) chemotrophy; (C) electrotrophy; (D) piezo-electrotrophy. While all microorganisms involve electron transfer in metabolism and can be broadly considered electroactive, this article narrowly defines electrotrophic microorganisms as those directly acquiring electrons from external sources (e.g., minerals or electrodes) to emphasize piezoelectrotrophy’s uniqueness—generating/acquiring electrical energy from mechanical forces via piezoelectric mechanisms. |
Here, we propose piezoelectrotrophy as a fourth major paradigm in microbial energetics, defined as the metabolic process enabling microorganisms to harness mechanical energy through piezoelectric mechanisms, converting it into electrical energy for cellular metabolism. This paradigm addresses a fundamental gap in our understanding: while mechanical energy is ubiquitous across all microbial habitats, from ocean sediments experiencing tidal forces to subsurface rocks subjected to tectonic stress, no established mechanisms exist for its direct utilization as a primary metabolic energy source. Unlike phototrophy (limited by light availability), chemotrophy (constrained by chemical gradients), and electrotrophy (requiring pre-existing electrical sources), piezoelectrotrophy could theoretically operate wherever mechanical force is present, potentially enabling microbial activity in environments previously considered too energy-poor to support metabolism.
While this review focuses primarily on prokaryotic microorganisms due to their predominance in extreme, energy-limited environments where piezoelectrotrophy may be most significant, the underlying principles could potentially extend to eukaryotic microorganisms such as protists, fungi, and microscopic algae. Future research should investigate piezoelectric capabilities across all microbial domains.
Mechanical energy represents one of the most ubiquitous forms of energy in the natural world, manifesting through geological processes, fluid dynamics, atmospheric movements, and countless other physical phenomena. From ocean wave motion to subtle vibrations caused by tectonic activity, mechanical energy is continuously generated and dissipated throughout Earth’s systems [1–3]. While light energy is continuously available across approximately half of Earth’s surface at any given time, it is restricted to surface environments and attenuates rapidly with depth in aquatic systems (>99% attenuation within 200 m in marine environments). Chemical energy gradients show spatial heterogeneity and can be locally depleted through microbial activity. Mechanical energy similarly shows temporal and spatial variation: tidal forces are periodic (12–24 h cycles), seismic activity is episodic, and bioturbation is localized. However, mechanical forces manifest across virtually all depth ranges, from surface sediments to deep subsurface lithosphere, including permanently dark environments where neither phototrophy nor photosynthetically-derived organic matter can directly support metabolism. This depth-independent distribution distinguishes mechanical energy as a potential metabolic resource.
The abundance of mechanical energy in natural systems presents a paradox: while omnipresent, no established biological mechanisms have been identified for its direct utilization by microorganisms as a primary metabolic energy source. Current mechanobiology understanding focuses primarily on mechanosensing and mechanotransduction, where mechanical forces serve as signals rather than energy sources [4]. Mechanosensitive ion channels respond to membrane tension, mechanoreceptors detect physical stimuli for sensory purposes, and mechanotransduction pathways regulate cellular processes in response to mechanical cues.
Recent advances in materials science have revealed the widespread occurrence of piezoelectric properties in both synthetic and natural materials [5,6]. Piezoelectricity, discovered in 1880 by Pierre and Jacques Curie, refers to the ability of certain materials to generate an electrical charge when subjected to mechanical force [7]. The phenomenon occurs in materials lacking a center of symmetry, where mechanical deformation causes displacement of positive and negative charge centers, resulting in electrical polarization. Piezoelectric effects have been documented in numerous minerals, including quartz, tourmaline, and topaz, as well as in biological materials such as collagen, chitin, and certain proteins.
The intersection of piezoelectric materials science with microbiology opens new possibilities for understanding how mechanical energy might be harnessed for biological processes. The conceptual framework of piezoelectrotrophy addresses several limitations inherent in existing paradigms. Where phototrophic strategies fail in dark environments, chemotrophic approaches are constrained by chemical availability, and electrotrophic mechanisms require specific conductive conditions [8], piezoelectrotrophy can potentially operate in any environment where mechanical force is present. The universality of mechanical forces suggests that piezoelectrotrophic strategies may enable microorganisms to harness supplementary energy in extreme environments, such as deep-sea sediments or subsurface lithospheres.
CONCEPTUAL FRAMEWORK: DEFINING A NEW FIELD
The establishment of piezoelectrotrophy as a distinct metabolic paradigm requires precise conceptual definitions and theoretical frameworks that distinguish it from related phenomena while integrating it within existing microbiological knowledge. Clear terminology and conceptual boundaries are essential for advancing scientific understanding and enabling productive research in the emerging field. Here, we first briefly introduce the three established energy-harvesting paradigms and subsequently define multiple terms and concepts involved in piezoelectrotrophy.
Three established energy-harvesting paradigms
Phototrophic microorganisms represent a well-established energy acquisition strategy, converting light energy into chemical energy through photosynthetic processes (Figure 1A). These organisms have evolved sophisticated light-harvesting complexes and electron transport chains that enable them to capture photons and convert light energy into electrochemical gradients. Specifically, electron transport chains generate proton gradients across membranes (photophosphorylation), which drive ATP synthesis through ATP synthase, while simultaneously producing reduced electron carriers such as NADPH in oxygenic processes or NADH in anoxygenic processes [9]. The phototrophic paradigm encompasses both oxygenic photosynthesis, performed by cyanobacteria, algae, and plants, and anoxygenic photosynthesis found in purple bacteria, green sulfur bacteria, green non-sulfur bacteria, and heliobacteria. Among prokaryotes, cyanobacteria are the primary oxygenic phototrophs and represent evolutionarily ancient lineages that fundamentally transformed Earth’s atmosphere. Although some phototrophic microorganisms have evolved low-light adaptation mechanisms, in completely dark environments, they must rely on organic carbon sources or enter dormancy states [10].
Chemotrophic microorganisms derive energy from chemical reactions, either through oxidation of organic compounds (chemoorganotrophy) or inorganic compounds (chemolithotrophy) (Figure 1B). These organisms have evolved highly diverse metabolic pathways that enable them to exploit a wide array of thermodynamically favorable chemical reactions available in nature [11]. From sulfur oxidation in deep-sea hydrothermal vents to methane oxidation in anaerobic sediments, chemotrophic microorganisms occupy ecological niches defined by specific chemical gradients and reaction potentials [12]. However, chemotrophic strategies remain constrained by the availability of suitable electron donors and acceptors, limiting their distribution in chemically homogeneous or depleted environments.
The most recently recognized paradigm, electrotrophy, involves direct uptake of electrons from external sources without traditional chemical mediators [13] (Figure 1C). Electrotrophic microorganisms can accept electrons from mineral surfaces, electrodes, or other microorganisms through specialized electron transport mechanisms, such as conductive pili with electron transfer capabilities [14–16]. These organisms, including species of Geobacter and Shewanella, as well as emerging examples in other genera like Rhodoferax, have evolved elaborate extracellular electron transfer networks that enable them to access energy sources previously considered biologically inaccessible [17–19]. While all microbial metabolism involves electron transfer, specialized extracellular electron transfer is limited to certain electroactive taxa [20]. In this review, we define electrotrophic microorganisms as those that directly acquire electrons from external insoluble sources (e.g., mineral surfaces or electrodes) to support growth and metabolism.
Definition of piezoelectrotrophy and its relationships with established paradigms
Piezoelectrotrophy is defined as the metabolic process whereby microorganisms convert mechanical energy into bioavailable electrical energy through interactions with piezoelectric materials or intrinsic piezoelectric biological structures. The process involves transduction of mechanical force, strain, or deformation into electrical potential, which is subsequently captured and utilized for cellular metabolic processes, including ATP synthesis, biosynthesis, and maintenance functions.
The definition encompasses several key mechanistic elements. First, the process requires a source of mechanical energy, which can be derived from environmental forces such as fluid flow, pressure changes, vibrations, or deformation. Second, the mechanism involves piezoelectric transduction, either through interaction with external piezoelectric materials or through intrinsic biological structures that exhibit piezoelectric properties. Third, the generated electrical energy must be captured and converted into metabolically useful forms through specialized cellular machinery (Figure 2A).
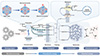 |
Figure 2 Hypothetical energy flow in piezoelectrotrophy. This schematic depicts the proposed transduction pathway for piezoelectrotrophy, converting mechanical energy (left input) to bioavailable chemical energy (right output) via nano-bio interface processes. Mechanical stress induces shape changes in piezoelectric materials, causing charge separation and the generation of electrons (e−) and protons (H+) through the piezoelectric effect. Captured charges transfer across the microbial membrane via conductive nanowires, outer membrane cytochromes, or soluble mediators, establishing a membrane potential and proton gradient. The membrane potential and proton gradient drive ATP synthesis by ATP synthase or the reduction of electron carriers like NAD(P)H, integrating with cellular metabolism for growth and maintenance. The mechanism distinguishes piezoelectrotrophy from established paradigms by utilizing ubiquitous mechanical forces in energy-limited environments. |
Piezoelectrotrophy differs fundamentally from mechanosensing or mechanotransduction in its primary function and energetic outcomes. While mechanosensitive processes respond to mechanical stimuli to regulate cellular behavior, piezoelectrotrophic processes harness mechanical energy to power cellular metabolism [21]. The energy capture aspect distinguishes piezoelectrotrophy from purely sensory mechanical responses and aligns it with other metabolic energy-harvesting strategies.
Temporal characteristics of piezoelectrotrophic processes may vary significantly depending on the mechanical energy source and cellular mechanisms involved. Some piezoelectrotrophic processes might operate continuously in environments with constant mechanical activity, while others might function intermittently in response to periodic mechanical disturbances. The ability to store captured electrical energy, either through cellular mechanisms or external capacitive systems, could enable sustained metabolism even during periods of reduced mechanical activity.
Piezoelectrotrophy exists within a broader landscape of microbial energy acquisition strategies and may interface with established paradigms in several ways. The relationship with electrotrophy is particularly close, as both involve electrical energy utilization, though the source differs: electrotrophy relies on pre-existing electrical gradients or conductors, while piezoelectrotrophy generates electrical energy de novo from mechanical force through piezoelectric transduction. Synergies with chemotrophy may occur in mechanically dynamic environments such as hydrothermal vents, where both chemical gradients and mechanical energy coexist. Microorganisms in these settings might employ mixed-mode metabolism, utilizing chemical energy during periods of substrate availability while supplementing with piezoelectrically-generated energy during mechanical disturbances. Integration with phototrophy is less direct but conceivable in shallow marine sediments where both light penetration and mechanical energy from wave action occur. These interconnections suggest that piezoelectrotrophy may function primarily as a supplementary energy source, enhancing metabolic flexibility rather than serving as a sole energy acquisition mode. This perspective aligns with the emerging understanding of metabolic plasticity in extreme environments.
Piezoelectrotrophic microorganisms
Piezoelectrotrophic microorganisms would possess specialized cellular structures, membrane adaptations, or symbiotic relationships that enable efficient capture and utilization of piezoelectrically generated electrical energy. Several groups of existing microorganisms possess characteristics that could predispose them to successful development or adaptation of piezoelectrotrophic capabilities [22].
Definition of electroactive microorganism
Electroactive microorganisms are prokaryotes and eukaryotes capable of transferring electrons to or from extracellular solid substrates, including minerals, electrodes, or other cells. These organisms employ several distinct mechanisms for extracellular electron transfer. Some species, such as Geobacter and Shewanella, use direct contact through outer membrane cytochromes. Geobacter sulfurreducens can also produce conductive protein nanowires or pili that facilitate electron transfer. Another strategy involves soluble electron shuttles, as demonstrated by Shewanella species that secrete flavins. Perhaps most remarkably, cable bacteria achieve long-distance electron conduction across significant spatial separations. These electroactive organisms inhabit diverse environments spanning sediments, soils, wastewater treatment systems, and hydrothermal systems. The phylogenetic breadth of this capability is substantial, encompassing at least 15 bacterial phyla along with some archaeal lineages, demonstrating that extracellular electron transfer has evolved across many branches of the microbial tree of life.
Three primary groups of microorganisms possess characteristics suggesting potential for piezoelectrotrophic adaptation: (1) electroactive bacteria of the families Geobacteraceae and Shewanellaceae, which already possess extracellular electron transfer mechanisms; (2) magnetotactic bacteria of the Magnetospirillum and Magnetococcus genera, which demonstrate sophisticated mineral interaction capabilities; and (3) cable bacteria of the family Desulfobulbaceae, which conduct electrons over centimeter distances through conductive filaments.
Electroactive microorganisms represent logical candidates for piezoelectrotrophic adaptation, as they already possess sophisticated extracellular electron transfer mechanisms that could potentially be modified to capture piezoelectric energy. Geobacter species produce conductive protein nanowires (pili) that enable electron transport over micrometer distances and could potentially interface with piezoelectric materials to capture generated electrical charge. These organisms also possess multiple outer membrane cytochromes that facilitate electron transfer across cellular boundaries and could serve as interfaces for piezoelectric energy capture.
Shewanella species exhibit metabolic flexibility and possess multiple electron transfer pathways, enabling them to utilize diverse electron acceptors. Their ability to reduce solid electron acceptors through direct contact and their production of electron transfer mediators suggest cellular capabilities that could be adapted for piezoelectric electron capture [23]. The production of flavins and other electron mediators by Shewanella species could facilitate electron capture from piezoelectric sources even in the absence of direct physical contact.
While magnetite is not piezoelectric, magnetotactic bacteria serve as an important biological precedent for microbe-mineral interactions. These organisms demonstrate the ability to biomineralize and interface with inorganic materials through specialized organelles called magnetosomes, showing that microorganisms can evolve sophisticated mechanisms to incorporate and utilize mineral particles for specific cellular functions. This biological precedent suggests that similar mineral-interfacing capabilities could potentially be adapted for interactions with piezoelectric materials, though the mechanisms would differ fundamentally due to the distinct physical properties of piezoelectric versus magnetic materials.
Recent experimental evidence suggests piezoelectrotrophic capabilities in several systems. Shewanella oneidensis MR-1 cultured on ZnO nanowire arrays showed enhanced electron transfer rates with a 2.3-fold increase under ultrasonic stimulation compared to static conditions, with corresponding increases in extracellular cytochrome expression [2]. In another study, mixed microbial communities from marine sediments demonstrated increased metabolic activity, measured by CO2 production, when cultivated with piezoelectric BaTiO3 particles subjected to periodic mechanical agitation [1]. Furthermore, electroactive biofilms of G. sulfurreducens exhibited enhanced current production with a 40% increase when grown on piezoelectric substrates under flow conditions compared to non-piezoelectric controls [24]. While these studies do not conclusively demonstrate piezoelectrotrophy as a primary metabolic strategy, they provide preliminary evidence for enhanced microbial activity associated with piezoelectric materials under mechanical stimulation.
Morphological and physiological characteristics of piezoelectrotrophic microorganisms would likely reflect their specialized energy-harvesting capabilities. Cell envelope modifications might include specialized membrane proteins designed to interface with piezoelectric materials, enhanced membrane conductivity to facilitate electrical energy capture, or modified membrane composition to optimize electrical properties [25]. Intracellular adaptations could involve modified electron transport chains that can accommodate variable electrical inputs, enhanced electrical storage capabilities through specialized organelles or membrane modifications, or altered metabolic regulation systems that respond to electrical energy availability.
Extension to eukaryotic microorganisms
While current evidence for piezoelectrotrophy derives mainly from studies on electroactive prokaryotes, eukaryotic microorganisms possess several characteristics that could support piezoelectrotrophic capabilities. Protists such as ciliates and flagellates generate substantial mechanical forces during locomotion and feeding, potentially enabling interaction with environmental piezoelectric materials. Diatoms possess silica frustules with demonstrated mechanical properties, and interactions between these structures and piezoelectric substrates could generate electrical energy that might be metabolically accessible. Fungi produce extensive hyphal networks that exert mechanical force during growth and can incorporate mineral particles into their structures; notably, some fungi are already known to interact with electrical fields, suggesting a pre-existing capacity for electrical sensing or utilization. Additionally, microalgae in biofilms experience continuous mechanical stress from fluid flow that could be harnessed through piezoelectric interfaces at the biofilm-substrate boundary.
These possibilities remain speculative and require experimental investigation, but they illustrate that piezoelectric energy transduction may represent a universal microbial capability rather than being restricted to prokaryotes. The mechanically active lifestyles of diverse eukaryotic microorganisms, combined with their frequent association with mineral surfaces in natural environments, provide multiple potential contexts where piezoelectrotrophic metabolism could emerge and provide a selective advantage (Table 1, [26–28]).
Candidate piezoelectrotrophic microorganisms and supporting evidence
Piezoelectromicrobiology as an emerging discipline
Piezoelectromicrobiology emerges as an interdisciplinary field that integrates microbiology, materials science, bioelectrochemistry, and mechanical engineering to study interactions between microorganisms and piezoelectric phenomena. The scope encompasses fundamental research into piezoelectrotrophic mechanisms, applied research for biotechnological applications, and ecological studies on mechanical energy utilization in natural systems. This interdisciplinary approach mirrors successful precedents in fields such as bioelectrochemistry and geomicrobiology, which have similarly integrated physical sciences with microbiology to reveal previously unrecognized microbial capabilities [29].
Primary objectives include the identification and characterization of naturally occurring piezoelectrotrophic microorganisms, elucidation of molecular mechanisms underlying piezoelectric energy capture and utilization, development of engineered piezoelectrotrophic systems for technological applications, and assessment of ecological roles and evolutionary significance of piezoelectrotrophic processes [18].
Methodological development represents a critical aspect, as standard microbiological techniques may prove insufficient for detecting and characterizing piezoelectrotrophic processes [30]. New approaches might include specialized cultivation techniques that incorporate mechanical stimulation, electrical measurement systems capable of detecting microbial piezoelectric interactions, and imaging technologies that can visualize mechanical-electrical transduction at the cellular level [31]. These methodological innovations will draw upon established techniques from bioelectrochemistry, electrochemical engineering, and nanoscale biophysics while adapting them to the unique requirements of piezoelectrotrophic research.
Theoretical mechanisms: Transduction pathways
The theoretical foundation of piezoelectrotrophy rests on plausible mechanisms for converting mechanical energy into forms that can support cellular metabolism. These mechanisms must account for physical processes of piezoelectric transduction, biological processes of energy capture and conversion, and integration of these processes within existing cellular metabolic networks.
Piezoelectric transduction represents the initial step in piezoelectrotrophic energy conversion, where mechanical force applied to piezoelectric materials generates electrical polarization due to charge separation in non-centrosymmetric materials [32]. The efficiency depends on the material’s piezoelectric coefficients, the magnitude and frequency of applied mechanical force, and the interface conditions between the piezoelectric material and the biological system (Figure 2B).
Electrical energy capture requires specialized biological mechanisms that can efficiently harvest the electrical potential generated through piezoelectric transduction. These mechanisms might involve modified electron transport proteins, specialized membrane channels, or bioelectrochemical pathways that convert electrical energy into reduced electron carriers [3,33]. Capture efficiency would depend on the electrical properties of the biological interface, the temporal characteristics of the electrical signal, and the capacity of cellular systems to store or immediately utilize captured energy.
Energy conversion represents the final step in piezoelectrotrophic metabolism, where captured electrical energy is transformed into conventional metabolic currencies such as ATP, reduced electron carriers, or proton gradients. Conversion might proceed through established bioelectrochemical pathways, such as those found in electrotrophic microorganisms, or through novel mechanisms specifically adapted for processing piezoelectrically generated electrical energy [34].
THE PIEZOELECTRIC-MICROBIAL INTERFACE: ELECTRON GENERATION, TRANSFER, AND UTILIZATION
The success of piezoelectrotrophic metabolism depends critically on the efficient operation of the nano-bio interface, where mechanical forces are converted to electrical energy and subsequently captured by microbial systems. Understanding the complete electron transport pathway, from initial generation through cellular utilization, requires examining each component process and its integration into a functional energy-harvesting system (Figure 3) [1,35-38].
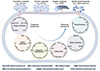 |
Figure 3 Material types and metabolic categories in representative piezo-electrotrophy studies. The references related to piezoelectric materials in the image include struvite [37], BaTiO3 [1], PVDF [35], and diphenylalanine [36]. The references related to metabolic processes in the image include carbon fixation [1,38], sulfur Reduction [1], organic degradation [1], cell growth [1], metal reduction, and denitrification [1,37]. |
Electron generation: Piezoelectric materials and mechanisms
Piezoelectricity refers to the generation of electric charge in response to mechanical force, or vice versa, due to the displacement of ions in non-centrosymmetric crystal lattices. The lack of inversion symmetry enables electromechanical coupling and occurs in 20 of 32 crystal point groups [39]. Recent advances in nanomaterials have amplified these properties through high surface-to-volume ratios, facilitating applications in energy harvesting and biomedicine.
Piezoelectric materials can be broadly classified into several categories based on their composition, structure, and origin.
(1) Crystalline materials, including quartz, tourmaline, and topaz, represent naturally occurring piezoelectric minerals that are abundant in many geological environments [28]. These materials exhibit well-characterized piezoelectric properties and have been extensively studied in materials science applications. Quartz demonstrates a piezoelectric coefficient d11 of approximately 2.3 pm/V and has been implicated in various geological phenomena, including potential electrical effects during earthquakes.
(2) Ceramic materials, such as zinc oxide (ZnO), barium titanate (BaTiO3), and lead-free alternatives, represent synthetic piezoelectric materials with enhanced piezoelectric coefficients compared to natural minerals. ZnO represents one of the most promising inorganic piezoelectric materials for biological applications due to its biocompatibility, environmental stability, and favorable piezoelectric properties [40]. ZnO nanowires exhibit piezoelectric coefficient d33 values of 12–26 pm/V, generating output voltages of 20–50 mV under physiologically relevant mechanical strains (0.1%–0.5%). These electrical potentials are within the range that can drive extracellular electron transfer in electroactive bacteria (10–500 mV). ZnO nanostructures can generate substantial electrical potentials when subjected to mechanical force while maintaining minimal cytotoxic effects. Barium titanate (BaTiO3) demonstrates higher piezoelectric coefficients (d33 ~ 191 pm/V) but poses biocompatibility concerns due to barium toxicity. Lead-free alternatives such as potassium sodium niobate (KNN) show promising d33 values of 80–416 pm/V with improved biocompatibility [40].
(3) Organic piezoelectric materials include biopolymers such as chitin (d14 ~ 0.2–2 pm/V) [5], the second most abundant natural polymer after cellulose, found in arthropod exoskeletons and fungal cell walls; cellulose (d14 ~ 0.1–1 pm/V) in plant cell walls; and polyvinylidene fluoride (PVDF) (d33 ~ 20–33 pm/V), a synthetic polymer with excellent biocompatibility and flexibility [35]. Collagen, the most abundant protein in animal tissues, exhibits piezoelectric properties that arise from its hierarchical structure and molecular asymmetry. Collagen demonstrates a piezoelectric coefficient d14 of approximately 0.1–2 pm/V, arising from its triple-helix structure and hierarchical organization [41]. When subjected to physiological mechanical loading (1–10 MPa stress during tissue deformation), collagen fibrils generate electrical potentials of 1–5 mV [42,43]. These potentials have been implicated in bone remodeling, where mechanical loading generates electrical signals that influence osteoblast and osteoclast activity. In microbial contexts, collagen-rich extracellular matrices in biofilms or sediments could theoretically serve as piezoelectric substrates for microbial energy harvesting, particularly in mechanically active environments such as intestinal mucosa or flowing aquatic systems. The ubiquity of collagen in animal tissues suggests evolutionary accessibility of piezoelectrotrophic mechanisms if selective pressure existed.
(4) Peptide-based piezoelectric materials include diphenylalanine, which self-assembles into highly ordered nanotubes with d15 coefficients of 17–60 pm/V, and designed peptide sequences incorporating β-sheet structures that enhance mechanical stability and piezoelectric response (d33 ~ 5–15 pm/V) [36]. The ability to genetically encode these peptides makes them particularly attractive for synthetic biology approaches to engineered piezoelectrotrophic systems. Short peptides with specific amino acid sequences can self-assemble into ordered structures that exhibit significant piezoelectric responses. The ability to design peptides with tailored properties enables optimization of piezoelectric performance for specific biological applications.
Electron transfer: From piezoelectric materials to microorganisms
Successful capture of piezoelectrically generated electrical energy by microbial systems requires efficient electron transfer mechanisms that bridge the gap between inorganic piezoelectric materials and biological cellular systems. The electron transfer process must overcome electrical, chemical, and physical barriers while maintaining biocompatibility and avoiding cellular damage.
Direct electron transfer mechanisms involve physical contact between cellular electron transport proteins and piezoelectric surfaces, enabling electron flow through protein-mediated pathways. Outer membrane cytochromes in bacteria such as Geobacter and Shewanella species can facilitate direct electron transfer across distances of several nanometers through protein-protein electron tunneling mechanisms [44].
Mediated electron transfer offers an alternative mechanism that can accommodate physical separation between piezoelectric materials and microbial cells through the action of soluble electron mediators. Small molecule mediators, including quinones, flavins, phenazines, and metal complexes, can shuttle electrons between piezoelectric sources and cellular electron transport chains [45].
Formation of conductive biofilm networks represents a community-level approach to extracellular electron transfer that could enhance the efficiency of piezoelectric energy capture. Biofilm communities can establish complex electrical networks that enable electron sharing and could potentially coordinate piezoelectric energy harvesting across multiple cells or species [46].
Electron utilization: Cellular integration of piezoelectric energy
Successful implementation of piezoelectrotrophy requires not only efficient electrical energy capture but also effective integration of captured energy with cellular metabolic processes. Utilization of piezoelectric energy must be compatible with existing cellular systems while providing sufficient energy flux to support essential cellular functions.
Conversion of captured electrical energy into metabolically useful forms requires specific biochemical pathways that interface electrical inputs with established cellular energy currencies. The most direct pathway involves conversion of electrical energy into ATP through chemiosmotic mechanisms, similar to those employed in respiration and photosynthesis.
Proton gradient establishment through electrical energy inputs could drive ATP synthesis through conventional ATP synthase mechanisms. Captured electrical energy could be used to drive proton pumping across cellular membranes, creating the electrochemical gradient necessary for ATP synthesis. Such pathways would integrate seamlessly with existing cellular bioenergetics and could provide substantial energy flux for cellular metabolism.
Alternative pathways for electrical energy utilization include direct reduction of biological electron acceptors such as NAD+, NADP+, or other electron carriers through electrically-driven electron transfer reactions. Reduction of electron carriers using captured electrical energy could support anabolic metabolism and provide a mechanism for storing electrical energy in chemical form [47].
Energy storage and buffering systems could enable sustained metabolism despite potentially variable piezoelectric energy inputs. These systems might involve enhanced membrane capacitance, modified metabolic regulation, or specialized storage compounds that accumulate during periods of high electrical energy availability and support metabolism during energy-limited periods.
ECOLOGICAL IMPLICATIONS AND TECHNOLOGICAL APPLICATIONS
The significance of piezoelectrotrophy extends beyond individual organismal metabolism to encompass broader ecological processes, biogeochemical cycles, and technological advances that could transform our understanding of microbial energy utilization and its applications in environmental and biomedical contexts.
Natural habitats for piezoelectrotrophic microbes
Marine sedimentary environments represent prime candidates for piezoelectrotrophic activity due to the combination of mechanical energy sources and abundance of piezoelectric materials. Ocean floor sediments experience constant mechanical force from water currents, tidal forces, and wave action, while containing significant quantities of quartz and other piezoelectric minerals. Pressure variations associated with deep-sea environments and mechanical forces generated by burrowing organisms could create additional sources of mechanical energy.
The complex three-dimensional structure of sedimentary matrices provides numerous interfaces where piezoelectric materials and microorganisms can interact. Grain-to-grain contacts in sediments create force concentration points where mechanical forces are amplified, potentially enhancing piezoelectric energy generation [48]. Porosity and permeability of sediments enable fluid flow and pressure transmission, which can generate sustained mechanical force on embedded piezoelectric materials.
Terrestrial soil environments offer similar opportunities for piezoelectrotrophic activity, with mechanical energy sources including plant root growth, soil settling, temperature-induced expansion and contraction, and water movement through soil pores. Mineral composition of soils often includes significant fractions of quartz and other piezoelectric materials, particularly in sandy soils and weathered rock formations [49].
Tectonically active regions present extreme examples of mechanically dynamic environments where piezoelectrotrophy might be particularly significant. Intense mechanical forces associated with fault movements, volcanic activity, and seismic events could generate substantial piezoelectric potentials in mineral-rich environments [50]. Unique microbial communities found in deep subsurface environments associated with tectonic activity might include piezoelectrotrophic organisms adapted to harness abundant mechanical energy in these settings (Figure 4A).
 |
Figure 4 Ecological implications and technological applications. (A) Speculative presence in extreme environments; (B) potential role in biogeochemical cycles and ecosystem functions: driving forces behind Earth’s matter cycling; (C) significance in dark, oligotrophic environments: new insights into microbial survival strategies; (D) coexistence with other energy harvesting strategies (mixed trophic systems); (E) bioelectronic devices powered by piezoelectrotrophic systems; (F) environmental sensing and monitoring applications; (G) bioremediation possibilities using mechanical energy in contaminated sites; (H) energy harvesting from ambient mechanical sources (vibrations, fluid flow, pressure). |
Biogeochemical cycles and ecosystem functions
Piezoelectrotrophic microorganisms could play previously unrecognized roles in biogeochemical cycling by coupling mechanical energy with chemical transformations in ways that are independent of traditional energy sources. The ability to harness mechanical energy could enable microbial processes to operate in environments and conditions where other energy sources are limited, potentially expanding the range of conditions under which biogeochemical processes occur (Figure 4B).
Carbon cycling processes could be influenced by piezoelectrotrophic mechanisms that enable microbial CO2 fixation through conversion of mechanical energy inputs. Piezoelectrotrophic carbon fixation could operate independently of light availability, potentially supporting primary productivity in dark environments and contributing to global carbon budgets in ways that are not currently recognized.
Nitrogen cycling processes, including nitrogen fixation, nitrification, and denitrification, require substantial energy inputs that are typically provided through chemical or light energy sources [51]. Piezoelectrotrophic capabilities could enable these processes to operate in environments where traditional energy sources are limited, potentially expanding the geographical and environmental range of nitrogen cycling processes.
Sulfur cycling, particularly in sedimentary and hydrothermal environments, could be enhanced by piezoelectrotrophic processes that couple mechanical energy with sulfur oxidation or reduction reactions [52]. The abundance of both mechanical energy and sulfur compounds in many geological environments suggests that piezoelectrotrophic sulfur cycling could be significant in these settings.
Metal cycling processes, including oxidation and reduction of iron, manganese, and other metals, could be driven by piezoelectrotrophic mechanisms that utilize mechanical energy to power electron transfer reactions. Coupling of mechanical force with metal redox chemistry could enable novel biogeochemical processes that operate independently of traditional energy sources.
Significance in energy-limited environments
Deep subsurface environments, characterized by the absence of light and limited chemical energy sources, represent challenging habitats where traditional metabolic strategies may be insufficient to support robust microbial communities. Potential availability of mechanical energy through geological processes could provide an alternative energy source, enabling microbial persistence and activity in these otherwise energy-limited environments (Figure 4C). Microorganisms may also establish mixed-mode energy acquisition systems through integrating piezoelectrotrophy with other strategies [53] (Figure 4D).
The deep biosphere, extending kilometers below Earth’s surface, harbors diverse microbial communities despite extreme limitations in traditional energy sources [54]. Recognition of mechanical energy as a potential metabolic resource could explain the abundance and activity of microorganisms in deep subsurface environments where chemical energy sources are scarce [55].
Abyssal ocean environments, characterized by perpetual darkness and limited nutrient inputs, might harbor piezoelectrotrophic communities that utilize mechanical energy generated by deep-sea currents, tidal forces, and hydrothermal circulation [56]. Constant mechanical activity of these environments could provide sustained energy sources for specialized microbial communities [57].
Polar environments, where low temperatures limit chemical reaction rates and seasonal light availability constrains photosynthetic activity, might rely on piezoelectrotrophic processes to maintain microbial activity during periods when other energy sources are inadequate [58]. Mechanical force generated by freeze-thaw cycles, ice movement, and permafrost dynamics could provide sustained energy sources for specialized microbial communities.
Technological applications: From concept to implementation
Principles of piezoelectrotrophy offer substantial potential for technological advancement across multiple sectors, from environmental monitoring to medical devices. Development of practical applications based on piezoelectrotrophic concepts requires integration of biological systems with engineering design principles to create functional hybrid technologies.
Bioelectronic devices
Integration of piezoelectrotrophic microorganisms with electronic components could create semi-self-sustaining bioelectronic devices that minimize reliance on external power sources, provided sufficient ambient mechanical energy is available (Figure 4E). These hybrid systems combine biological energy-harvesting with electronic processing and communication, enabling autonomous operation in remote environments [6]. Sensor networks powered by piezoelectrotrophic systems could provide long-term monitoring without battery replacement, harvesting energy from wind, water flow, or vibrations [59]. Communication devices could maintain connectivity in disaster areas or remote stations, while low-power computing supports data logging or control functions, similar to microbial fuel cells [60]. Hybrid interfaces enable bidirectional communication via electrical signals, though challenges include signal latency and impedance matching [61]. Biocompatible components must avoid toxicity, ion leaching, and pH disruption to preserve microbial metabolism.
Environmental sensing and monitoring
Piezoelectrotrophic systems excel in environmental sensing where traditional power is impractical, with energy conversion efficiencies of 5%–10% under optimal stress. Seismic monitoring could convert geological stresses into electrochemical potentials via piezoelectric nanomaterials, powering early warning systems for earthquakes or eruptions, limited by transient bursts (Figure 4F). Structural health monitoring for buildings or bridges uses vibrations to detect damage or corrosion, enabling predictive maintenance [62]. Water quality systems harness flow turbulence to power sensors for contaminants or pH, operating continuously in rivers or groundwater. Atmospheric stations use wind for air quality data, supporting dense networks [63]. Wildlife monitoring employs animal movements to power tracking devices, minimizing disturbance [64].
Bioremediation applications
Contaminated sites often feature mechanical energy that piezoelectrotrophic mechanisms can harness for bioremediation, enhancing kinetics via electric fields (Figure 4G). Groundwater systems use flow to drive degradation of organic pollutants or metal reduction through enhanced electron transfer [65]. Sediment remediation leverages tidal action or disturbances for mixing and energy, increasing contaminant bioavailability [66]. Soil applications utilize freeze-thaw cycles or root growth for distributed treatment across large areas, without external inputs. Waste processing captures energy from mixing or aeration to improve efficiency [27]. In situ systems enable continuous operation using natural mechanical sources, gradually reducing contaminants toward equilibrium [67].
Energy harvesting from ambient mechanical sources
Ambient mechanical energy from vibrations, flow, and pressure enables piezoelectrotrophic harvesting via flexible, stable materials, minimizing environmental impact (Figure 4H). Transportation systems capture traffic vibrations to power roadside sensors or smart networks, reducing fossil fuel reliance. Building vibrations from occupants or wind support automation or controls, using robust composites against pollutants [68]. Industrial operations require power monitoring equipment, though corrosion-resistant designs are needed. Natural sources like wind or geological processes provide reliable power for remote instrumentation, with multi-source harvesting for variable conditions.
Implantable medical devices
The body generates mechanical energy through heartbeat, breathing, and movements, harvestable by piezoelectrotrophic systems to power implants, eliminating battery replacements if efficiencies exceed 1% and biocompatibility minimizes inflammation [68]. Cardiac pacemakers use contractions for stimulation, reducing surgical risks. Glucose monitors for diabetes harness body movements with enzymatic interfaces for real-time data. Neural implants for Parkinson’s or pain use movements for stimulation, enabling sophisticated therapy. Drug delivery systems power pumps for precise release [69]. Orthopedic implants monitor healing under loading, detecting complications via osteoblast responses [70]. Diagnostic sensors track blood chemistry or inflammation, providing physiological insights into energy balance demand.
RESEARCH CHALLENGES AND FUTURE DIRECTIONS
Advancement of piezoelectromicrobiology as a scientific discipline necessitates systematic investigation of fundamental questions, development of specialized methodologies, and validation of theoretical concepts through experimental evidence. A comprehensive research roadmap must address both basic science questions and practical implementation challenges.
Methodological challenges in detecting and measuring piezoelectrotrophy
Detection and quantification of piezoelectrotrophic processes pose unique methodological challenges that demand specialized experimental approaches and instrumentation to capture subtle electrical signals generated from microbial piezoelectric interactions. Traditional microbiological methods are often insufficient for resolving these electrical phenomena, requiring adaptation and development of analytical techniques that integrate electromechanical sensing capabilities.
Electrical measurement techniques for detecting microbial piezoelectric interactions must function at spatial scales aligned with cellular dimensions while offering sensitivity to biologically relevant signals. To date, these techniques have been applied only in preliminary or indirect ways to piezoelectrotrophic questions.
Microelectrode arrays have been used to measure electrical activity in electroactive biofilms, but have not yet been deployed specifically to detect piezoelectrically-generated potentials in microbial systems. Scanning probe microscopy techniques, including conductive AFM and Kelvin probe force microscopy, have characterized piezoelectric properties of biomaterials and microbial nanowires separately, but the combined application to detect microbial piezoelectric energy capture remains unexplored [71]. Similarly, impedance spectroscopy has detected electrical changes in microbial cultures but has not been adapted to distinguish piezoelectrically-derived signals from other electrical phenomena.
Development and optimization of these techniques specifically for piezoelectrotrophic studies represents an immediate methodological priority. Multiple challenges must be addressed systematically, including signal-to-noise ratios where biological signals of approximately 1–100 mV must be detected amid noise levels around 10 mV, the biocompatibility of electrode materials, and the disambiguation of piezoelectric effects from other electrochemical processes occurring simultaneously in microbial systems [72].
Specific methodological protocols for piezoelectrotrophic cultivation involve mechanical stimulation systems that incorporate several key components working in concert. Ultrasonic actuation at frequencies of 20–100 kHz with controlled intensity ranging from 0.1 to 2 W/cm2 is essential to generate reproducible mechanical stress on piezoelectric materials [73,74]. These systems should also include microfluidic devices with integrated piezoelectric elements subjected to periodic flow at 0.1–10 Hz, which effectively mimics tidal or peristaltic forces in biological environments [75]. Additionally, piezoelectric bioreactors with embedded ZnO or BaTiO3 nanostructures must be subjected to controlled compression cycles operating at 0.1–1 Hz with pressures of 10–100 kPa [2,76]. To monitor the electrical activity generated by these mechanical perturbations, in situ electrical monitoring using microelectrode arrays with 10–100 μm electrode spacing enables real-time measurement of generated potentials and current in the nA–μA range [8,77].
Rigorous experimental validation requires three essential control conditions. First, mechanically stimulated non-piezoelectric materials must be tested to isolate the piezoelectric contribution from purely mechanical effects. Second, piezoelectric materials without mechanical stimulation serve as baseline controls to confirm that electrical generation requires mechanical input. Third, biological samples without piezoelectric materials under mechanical stimulation help distinguish between direct mechanical effects on cells and piezoelectrically mediated responses.
Cultivation methods for piezoelectrotrophic microorganisms should incorporate mechanical stimulation systems capable of generating controlled, repeatable force patterns. These systems must integrate seamlessly with standard microbiological techniques while enabling precise control over mechanical energy inputs. Developing specialized bioreactors that combine mechanical actuators with in situ electrical monitoring is essential for studying these organisms under realistic conditions.
Separating and purifying piezoelectrotrophic microorganisms from complex environmental samples demands techniques that differentiate them based on unique electromechanical properties within diverse microbial communities. Electrical activity-based methods, including dielectrophoresis and impedance spectroscopy sorting, hold promise for isolating electrically responsive cells, though challenges in specificity must be overcome.
Key questions regarding efficiency and ecological relevance
Ecological significance of piezoelectrotrophy hinges on its energy conversion efficiency and achievable energy fluxes through piezoelectric mechanisms. Addressing fundamental questions on efficiency and ecological relevance is essential to assess the potential role of piezoelectrotrophic processes in natural systems.
Comparative energy flux analysis to assess the plausible contribution of piezoelectrotrophy to microbial energetics involves comparing typical energy fluxes across established metabolic paradigms.
Phototrophy operates with solar irradiance at Earth’s surface averaging approximately 1000 W/m2 under optimal conditions, with phototrophic organisms achieving 1%–6% conversion efficiency. Such conversion efficiency yields 10–60 W/m2 of captured energy, representing one of the most productive energy capture systems in nature [78]. Chemotrophy shows highly variable energy flux from chemical reactions, ranging from 0.001 to 100 W/m2 depending on substrate availability and reaction kinetics. In specialized environments like deep-sea hydrothermal vents, chemical energy flux can reach 10–50 W/m2[79] supporting robust microbial communities despite the absence of light. Electrotrophy, a more recently characterized metabolic mode, demonstrates reported energy fluxes ranging from 0.01 to 1 W/m2 in natural sediments [80] to 10–100 W/m2 in optimized bioelectrochemical systems [81]. These values represent an intermediate energy availability between low-flux chemotrophic systems and high-productivity phototrophic environments. Mechanical energy availability presents a more complex picture. While ocean wave motion generates 2–3 kW/m2 of coastline, only a fraction of this energy is accessible at microbial scales. Sediment deformation from tidal forces or bioturbation might yield 0.001–0.1 W/m2 of localized mechanical energy [82], representing the potential substrate for piezoelectrotrophic metabolism.
For piezoelectrotrophy to achieve ecological significance, conversion efficiency would need to reach at least 1%–5% to generate energy fluxes of 0.00001–0.005 W/m2, assuming mechanical energy availability of 0.001–0.1 W/m2. While these fluxes appear modest compared to phototrophy or optimal chemotrophy, they could be sufficient for slow-growing microorganisms in energy-limited environments where other energy sources are scarce or absent. This efficiency threshold represents a critical experimental target for validation studies and defines the boundary conditions under which piezoelectrotrophy might confer a selective advantage.
Quantifying piezoelectric power flux requires integrated measurements:
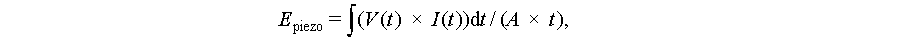
where V(t) is time-dependent voltage, I(t) is current, A is surface area, and t is time (t represents the integration period, chosen based on the characteristic timescale of the mechanical stimulus, ensuring it captures representative energy flux without introducing artifacts from transient effects). This calculation yields the average power density in W/m2, representing the rate of energy flux available for microbial utilization.
Conversion efficiency (η) can be assessed as
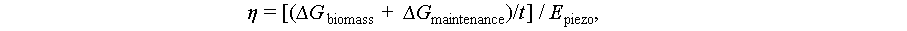
where ΔGbiomass represents free energy stored in new biomass and ΔGmaintenance covers maintenance metabolism, and t is time. Efficiency values >1% would indicate a significant piezoelectrotrophic contribution to cellular energetics, comparable to reported electrotrophic efficiencies.
Questions on energy conversion efficiency center on the fraction of mechanical energy captured and transformed into metabolically useful forms through piezoelectrotrophic pathways. Efficiency is influenced by factors including the piezoelectric material’s coupling coefficient, bio-material interface properties, microbial electron capture mechanisms, and downstream metabolic efficiencies. Theoretical modeling and empirical studies dissecting each step are essential to quantify overall system performance and identify bottlenecks.
Ecological energy flux inquiries examine whether piezoelectrotrophic processes yield adequate energy to sustain meaningful biological activity in nature. Benchmarking these fluxes against established sources like chemolithotrophy or photosynthesis is crucial to evaluate the contribution of mechanical energy in microbial ecosystems.
Questions on environmental abundance probe the prevalence of piezoelectric materials and mechanical energy sources in natural settings. Comprehensive surveys quantifying mineral distributions, energy generation rates, and favorable conditions are essential to identify ecological niches for piezoelectrotrophy.
Inquiries into competition and coexistence explore interactions between piezoelectrotrophic microbes and community counterparts, including resource partitioning or syntrophic relationships with alternative energy strategies. Understanding these dynamics is essential for predicting scenarios where piezoelectrotrophy provides selective advantages.
Critical experiments for validation
Validating piezoelectrotrophy as a viable metabolic strategy demands rigorously designed experiments yielding unequivocal evidence for microbial harnessing of mechanically derived electrical energy. These must differentiate piezoelectrotrophic mechanisms from confounding electrical effects while establishing definitive metabolic advantages.
Proof-of-concept experiments should establish the fundamental viability of piezoelectrotrophic energy capture by demonstrating microbial metabolic gains from solely mechanically sourced electricity. Such setups could employ isolated systems with mechanical input as the exclusive energy form, monitoring biological metrics like respiration rates or gene expression, alongside negative controls lacking mechanical stimulation to confirm causality [83].
Mechanistic validation experiments must delineate precise molecular and cellular pathways for piezoelectrotrophic energy capture and utilization. These could encompass detailed profiling of charge transport, proteomics for identifying key proteins, and demonstration of conversion to metabolic currencies like NADH/ATP.
Environmental validation experiments need to confirm piezoelectrotrophic activity in situ and quantify its ecological impact. These might include field investigations in predicted hotspots like sedimentary environments, integrated with laboratory recreations using environmental samples to validate observations.
Comparative analyses should evaluate the efficiency and competitive advantage of piezoelectrotrophy against alternatives like chemotrophy or phototrophy. Such comparative studies could entail head-to-head assessments of growth kinetics, thermodynamic efficiencies, and fitness in co-cultures under diverse conditions.
Future directions and technological development
Future development of piezoelectromicrobiology will likely involve increasing integration with other emerging technologies and expansion into new application domains that leverage the unique capabilities of mechanically-powered biological systems.
Synthetic biology integration offers robust tools for engineering and enhancing piezoelectrotrophic traits. Genetic engineering strategies could retrofit existing microbes with piezoelectrotrophic functions or amplify electrical energy pathways [84]. Protein engineering methods could craft bespoke electron carriers, bio-interface proteins, or conversion enzymes tuned for piezoelectric contexts [85].
Engineered hybrid systems fusing biological and synthetic elements could yield piezoelectrotrophic platforms with superior performance metrics compared to standalone biological or synthetic counterparts. These amalgamations would exploit biological adaptability and synthetic precision to offset individual component limitations.
Advanced materials development could significantly enhance piezoelectrotrophic system performance through improved piezoelectric materials, better bio-material interfaces, and novel hybrid materials that combine biological and synthetic components. Nanoscale materials engineering could optimize surface properties for biological interaction while maintaining high energy conversion efficiency.
Principles of piezoelectrotrophy might extend to complex biological entities, such as multicellular organisms or tissues, advancing energy biology and biotechnology. Plant architectures incorporating piezoelectric harvesting could exploit wind-driven forces to augment photosynthesis. Animal frameworks endowed with piezoelectrotrophic traits could reduce metabolic demands through capturing forces from locomotion [86].
Confluence of piezoelectromicrobiology with nascent biotechnologies fosters synergistic advancements. Bioelectronics integration could combine piezoelectrotrophic energy harvesting with biological computing and information processing capabilities. Nanobiotechnology convergence could enable the development of nanoscale piezoelectrotrophic systems with unique properties and applications.
CONCLUSION
Piezoelectrotrophy represents an emerging paradigm in microbial energetics that expands energy acquisition strategies available to microorganisms. The concept builds upon established principles, including charge separation in asymmetric materials, extracellular electron transfer mechanisms in electroactive bacteria, and bioenergetic pathways that couple electron transport to ATP synthesis. Through piezoelectric conversion of mechanical stress into electrical potential, microorganisms could theoretically harness ubiquitous mechanical forces as an energy source. The framework connects physical and biological processes, potentially addressing energy limitations in environments where light and chemical energy sources are scarce.
Ecological implications of piezoelectrotrophy extend across multiple scales and environments. In extreme habitats where conventional phototrophic, chemotrophic, or electrotrophic strategies prove insufficient, mechanical energy conversion could enable microbial survival and persistence. Such capabilities would require fundamental revisions to current biogeochemical models and our understanding of ecosystem dynamics. From an evolutionary perspective, mechanical energy may have supported early life forms in environments rich in piezoelectric minerals and characterized by frequent tectonic activity. These conditions could have enhanced mechano-chemical nutrient cycles during early Earth history.
Practical significance and future applications of piezoelectrotrophy involve validation that carries significant practical implications for sustainable biotechnology. In climate-impacted environments—including thawing permafrost, degraded soils, and oxygen-depleted coastal zones—piezoelectrotrophic systems could enable bioremediation powered solely by ambient mechanical energy (wind, water flow, or freeze-thaw cycles), eliminating dependence on external energy inputs or chemical additions. Self-powered biosensors based on piezoelectrotrophic principles could monitor remote or hostile environments (deep ocean, subsurface, polar regions) without battery replacement, supporting long-term environmental assessment critical for climate change response.
Beyond terrestrial applications, piezoelectrotrophic capabilities expand the potential habitability envelope for extraterrestrial life detection. Enceladus’s subsurface ocean experiences continuous tidal flexing, generating mechanical energy, while Mars’s subsurface may harbor piezoelectric minerals subjected to seismic activity. Piezoelectrotrophic metabolism could potentially support microbial communities in such mechanically active but light-absent and chemically limited environments, informing astrobiology search strategies and life detection instrumentation design. These applications underscore the practical value of pursuing piezoelectrotrophy research beyond fundamental science.
Current research faces significant challenges in efficiency optimization, detection methods, and experimental validation. Interdisciplinary approaches are essential for accurate quantification of energy conversion rates and demonstration of piezoelectrotrophy under natural conditions. Rigorous empirical testing remains necessary to establish the viability and ecological relevance of mechanical energy utilization by microorganisms. Successful validation of piezoelectrotrophy could fundamentally alter our understanding of microbial resilience and expand boundaries between biological and physical processes. Such advances would create new opportunities for biotechnological applications in energy-limited environments, with potential implications for both terrestrial ecosystem management and extraterrestrial life detection strategies.
Funding
This study was supported by the National Natural Science Foundation of China (42525702, 42307466, 42577284, 42307176).
Conflict of interest
The authors declare no conflict of interest.
References
- Ren G, Ye J, Liu L, et al. Mechanical energy drives the growth and carbon fixation of electroactive microorganisms. Engineering 2025; 47: 194-203. [Article] [Google Scholar]
- Guo M, Li Y, Qiao S. Bacteria-piezocatalyst for NO3− reduction to NH4+ driven by hydraulic kinetic energy. Water Res 2025; 286: 124292. [Article] [Google Scholar]
- Ren G, Ye J, Hu Q, et al. Growth of electroautotrophic microorganisms using hydrovoltaic energy through natural water evaporation. Nat Commun 2024; 15: 4992. [Article] [Google Scholar]
- Di X, Gao X, Peng L, et al. Cellular mechanotransduction in health and diseases: From molecular mechanism to therapeutic targets. Sig Transduct Target Ther 2023; 8: 282. [Article] [Google Scholar]
- Wang R, Sui J, Wang X. Natural piezoelectric biomaterials: A biocompatible and sustainable building block for biomedical devices. ACS Nano 2022; 16: 17708-17728. [Article] [Google Scholar]
- Chen S, Tong X, Huo Y, et al. Piezoelectric biomaterials inspired by nature for applications in biomedicine and nanotechnology. Adv Mater 2024; 36: 2406192. [Article] [Google Scholar]
- Curie JC, Curie P. Development, via compression, of electric polarization in hemihedral crystals with inclined faces. Bull Soc Minerol France 1880; 3: 90–93 [Google Scholar]
- Lovley DR, Holmes DE. Electromicrobiology: The ecophysiology of phylogenetically diverse electroactive microorganisms. Nat Rev Microbiol 2022; 20: 5-19. [Article] [Google Scholar]
- Blankenship RE. Molecular Mechanisms of Photosynthesis, 3rd ed. Hoboken: Wiley-Blackwell, 2021 [Google Scholar]
- Rajeev L, da Rocha UN, Klitgord N, et al. Dynamic cyanobacterial response to hydration and dehydration in a desert biological soil crust. ISME J 2013; 7: 2178-2191. [Article] [Google Scholar]
- Madigan MT, Aiyer J, Buckley DH, et al. Brock Biology of Microorganisms, 16th ed. Boston: Pearson, 2021 [Google Scholar]
- Dai Z, Fan Y, Li Y, et al. Global patterns and environmental drives of nitrite/nitrate-dependent anaerobic methane oxidation across natural ecosystems. Glob Change Biol 2025; 31: e70350. [Article] [Google Scholar]
- Zhang JZ, Sokol KP, Paul N, et al. Competing charge transfer pathways at the photosystem II-electrode interface. Nat Chem Biol 2016; 12: 1046-1052. [Article] [Google Scholar]
- Reguera G, McCarthy KD, Mehta T, et al. Extracellular electron transfer via microbial nanowires. Nature 2005; 435: 1098-1101. [Article] [Google Scholar]
- Malvankar NS, Vargas M, Nevin KP, et al. Tunable metallic-like conductivity in microbial nanowire networks. Nat Nanotech 2011; 6: 573-579. [Article] [Google Scholar]
- El-Naggar MY, Wanger G, Leung KM, et al. Electrical transport along bacterial nanowires from Shewanella oneidensis MR-1. Proc Natl Acad Sci USA 2010; 107: 18127-18131. [Article] [Google Scholar]
- Kotloski NJ, Gralnick JA, Newman DK. Flavin electron shuttles dominate extracellular electron transfer by Shewanella oneidensis. mBio 2013; 4: e00553-12. [Article] [Google Scholar]
- Nealson KH, Rowe AR. Electromicrobiology: Realities, grand challenges, goals and predictions. Microb Biotechnol 2016; 9: 595-600. [Article] [Google Scholar]
- Gralnick JA, Bond DR. Electron transfer beyond the outer membrane: Putting electrons to rest. Annu Rev Microbiol 2023; 77: 517-539. [Article] [Google Scholar]
- Digel L, Bonné R, Aiyer K. Are all microbes electroactive?. Cell Rep Phys Sci 2024; 5: 102200. [Article] [Google Scholar]
- Young MN, Sindoni MJ, Lewis AH, et al. The energetics of rapid cellular mechanotransduction. Proc Natl Acad Sci USA 2023; 120: e2215747120. [Article] [Google Scholar]
- Baker IR, Conley BE, Gralnick JA, et al. Evidence for horizontal and vertical transmission of mtr-mediated extracellular electron transfer among the bacteria. mBio 2022; 13: e02904. [Article] [Google Scholar]
- He X, Lu H, Fu J, et al. Promotion of direct electron transfer between Shewanella putrefaciens CN32 and carbon fiber electrodes via in situ growth of α-Fe2O3 nanoarray. Front Microbiol 2024; 15: 1407800. [Article] [Google Scholar]
- Chen M, Zhou X, Liu X, et al. Facilitated extracellular electron transfer of Geobacter sulfurreducens biofilm with in situ formed gold nanoparticles. Biosens Bioelectron 2018; 108: 20-26. [Article] [Google Scholar]
- Zhao J, Li F, Cao Y, et al. Microbial extracellular electron transfer and strategies for engineering electroactive microorganisms. Biotechnol Adv 2021; 53: 107682. [Article] [Google Scholar]
- Chen L, An X, Zhao S, et al. Multienergy codriven electron transfer across the nano-bio interface for efficient photobiocatalysis. ACS Nano 2025; 19: 11164-11175. [Article] [Google Scholar]
- Liu W, Fu P, Zhang Y, et al. Efficient hydrogen production from wastewater remediation by piezoelectricity coupling advanced oxidation processes. Proc Natl Acad Sci USA 2023; 120: e2218813120. [Article] [Google Scholar]
- Voisey CR, Hunter NJR, Tomkins AG, et al. Gold nugget formation from earthquake-induced piezoelectricity in quartz. Nat Geosci 2024; 17: 920-925. [Article] [Google Scholar]
- Gadd GM. New horizons in geomycology. Environ Microbiol Rep 2017; 9: 4-7. [Article] [Google Scholar]
- Bedendi G, De Moura Torquato LD, Webb S, et al. Enzymatic and microbial electrochemistry: Approaches and methods. ACS Meas Sci Au 2022; 2: 517-541. [Article] [Google Scholar]
- Fernandes MM, Carvalho EO, Lanceros-Mendez S. Electroactive smart materials: Novel tools for tailoring bacteria behavior and fight antimicrobial resistance. Front Bioeng Biotechnol 2019; 7: 277. [Article] [Google Scholar]
- Kim JH, Yoon H, Viswanath S, et al. Conformable piezoelectric devices and systems for advanced wearable and implantable biomedical applications. Annu Rev BioMed Eng 2025; 27: 255-282. [Article] [Google Scholar]
- Kracke F, Vassilev I, Krömer JO. Microbial electron transport and energy conservation—The foundation for optimizing bioelectrochemical systems. Front Microbiol 2015; 6: 575. [Article] [Google Scholar]
- Kim B, Baek G, Kim C, et al. Progress and prospects for applications of extracellular electron transport mechanism in environmental biotechnology. ACS EST Eng 2024; 4: 1520-1539. [Article] [Google Scholar]
- Bera B, Sarkar MD. Piezoelectricity in PVDF and PVDF based piezoelectric nanogenerator: A concept. IOSR JAP 2017; 09: 95-99. [Article] [Google Scholar]
- Yuan H, Han P, Tao K, et al. Piezoelectric peptide and metabolite materials. Research 2019; 2019: 2019/9025939. [Article] [Google Scholar]
- Ye J, Ren G, Liu L, et al. Wastewater denitrification driven by mechanical energy through cellular piezo-sensitization. Nat Water 2024; 2: 531-540. [Article] [Google Scholar]
- Tremblay PL, Xu M, Joya MB, et al. A biopiezocatalyst harnessing mechanical energy to enhance bioplastic production from CO2 and organic carbon. Nat Commun 2025; 16: 8141. [Article] [Google Scholar]
- He Q, Briscoe J. Piezoelectric energy harvester technologies: Synthesis, mechanisms, and multifunctional applications. ACS Appl Mater Interfaces 2024; 16: 29491-29520. [Article] [Google Scholar]
- Chen B, Feng Z, Yao FZ, et al. Flexible piezoelectrics: Integration of sensing, actuating and energy harvesting. npj Flex Electron 2025; 9: 58. [Article] [Google Scholar]
- Zhang Y, Wang X, Wang W, et al. Unlocking piezoelectric potential in collagen: Intrafibrillar mineralization matters. ACS Nano 2025; 19: 26411-26424. [Article] [Google Scholar]
- Denning D, Kilpatrick JI, Fukada E, et al. Piezoelectric tensor of collagen fibrils determined at the nanoscale. ACS BioMater Sci Eng 2017; 3: 929-935. [Article] [Google Scholar]
- Fernandez-Yague MA, Trotier A, Demir S, et al. A self-powered piezo-bioelectric device regulates tendon repair-associated signaling pathways through modulation of mechanosensitive ion channels. Adv Mater 2021; 33: 2008788. [Article] [Google Scholar]
- Hou L, Cortez R, Hagerman M, et al. Co-occurrence of direct and indirect extracellular electron transfer mechanisms during electroactive respiration in a dissimilatory sulfate reducing bacterium. Microbiol Spectr 2025; 13: e01226-24. [Article] [Google Scholar]
- Shaw DR, Katuri KP, Sapireddy V, et al. Independently evolved extracellular electron transfer pathways in ecologically diverse Desulfobacterota. ISME J 2025; 19: wraf097. [Article] [Google Scholar]
- Neu J, Shipps CC, Guberman-Pfeffer MJ, et al. Microbial biofilms as living photoconductors due to ultrafast electron transfer in cytochrome OmcS nanowires. Nat Commun 2022; 13: 5150. [Article] [Google Scholar]
- Choi O, Kim T, Woo HM, et al. Electricity-driven metabolic shift through direct electron uptake by electroactive heterotroph Clostridiumpasteurianum. Sci Rep 2014; 4: 6961. [Article] [Google Scholar]
- Hassenkam T, Skovbjerg LL, Stipp SLS. Probing the intrinsically oil-wet surfaces of pores in North Sea chalk at subpore resolution. Proc Natl Acad Sci USA 2009; 106: 6071-6076. [Article] [Google Scholar]
- Jiang YB, Zhong WH, Han C, et al. Characterization of electricity generated by soil in microbial fuel cells and the isolation of soil source exoelectrogenic bacteria. Front Microbiol 2016; 7: 1776. [Article] [Google Scholar]
- Stone J, Edgar JO, Gould JA, et al. Tectonically-driven oxidant production in the hot biosphere. Nat Commun 2022; 13: 4529. [Article] [Google Scholar]
- Ortiz M. All-inclusive nitrifiers in Antarctic soils. Nat Commun 2024; 15: 3144. [Article] [Google Scholar]
- Flieder M, Buongiorno J, Herbold CW, et al. Novel taxa of Acidobacteriota implicated in seafloor sulfur cycling. ISME J 2021; 15: 3159-3180. [Article] [Google Scholar]
- Tsoi R, Wu F, Zhang C, et al. Metabolic division of labor in microbial systems. Proc Natl Acad Sci USA 2018; 115: 2526-2531. [Article] [Google Scholar]
- Colman DR, Poudel S, Stamps BW, et al. The deep, hot biosphere: Twenty-five years of retrospection. Proc Natl Acad Sci USA 2017; 114: 6895-6903. [Article] [Google Scholar]
- Steele A, Benning LG, Wirth R, et al. Organic synthesis associated with serpentinization and carbonation on early Mars. Science 2022; 375: 172-177. [Article] [Google Scholar]
- D’Hondt S, Pockalny R, Fulfer VM, et al. Subseafloor life and its biogeochemical impacts. Nat Commun 2019; 10: 3519. [Article] [Google Scholar]
- Lathe R. Tidal chain reaction and the origin of replicating biopolymers. Int J AstroBiol 2005; 4: 19-31. [Article] [Google Scholar]
- Chen S, Gu Y, Liu E, et al. Freeze-thaw strength increases microbial stability to enhance diversity-soil multifunctionality relationship. Commun Earth Environ 2024; 5: 578. [Article] [Google Scholar]
- Singh D, Saurabh S, Li P, et al. Piezoelectric energy harvesting for pacemaker applications: Current state-of-the-art, materials, design, and alternative technologies. ACS BioMater Sci Eng 2025; 11: 4570-4620. [Article] [Google Scholar]
- Kang M, Lee DM, Hyun I, et al. Advances in bioresorbable triboelectric nanogenerators. Chem Rev 2023; 123: 11559-11618. [Article] [Google Scholar]
- Ouyang H, Liu Z, Li N, et al. Symbiotic cardiac pacemaker. Nat Commun 2019; 10: 1821. [Article] [Google Scholar]
- Li Z, Roscow J, Khanbareh H, et al. Energy harvesting from water flow by using piezoelectric materials. Adv Energy Sustain Res 2024; 5: 2300235. [Article] [Google Scholar]
- Lu ZQ, Zhao L, Fu HL, et al. Ocean wave energy harvesting with high energy density and self-powered monitoring system. Nat Commun 2024; 15: 6513. [Article] [Google Scholar]
- Li H, Tian C, Lu J, et al. An energy harvesting underwater acoustic transmitter for aquatic animals. Sci Rep 2016; 6: 33804. [Article] [Google Scholar]
- Ran M, Du B, Liu W, et al. Dynamic defects boost in-situ H2O2 piezocatalysis for water cleanup. Proc Natl Acad Sci USA 2024; 121: e2317435121. [Article] [Google Scholar]
- Kewalramani JA, Wang B, Marsh RW, et al. Coupled high and low-frequency ultrasound remediation of PFAS-contaminated soils. Ultrasons SonoChem 2022; 88: 106063. [Article] [Google Scholar]
- Romantschuk M, Lahti-Leikas K, Kontro M, et al. Bioremediation of contaminated soil and groundwater by in situ biostimulation. Front Microbiol 2023; 14: 1258148. [Article] [Google Scholar]
- Dagdeviren C, Yang BD, Su Y, et al. Conformal piezoelectric energy harvesting and storage from motions of the heart, lung, and diaphragm. Proc Natl Acad Sci USA 2014; 111: 1927-1932. [Article] [Google Scholar]
- Golafshan N, Patra S, Huynh ND, et al. Self-powered implantable and ingestible devices: Harvesting energy within the body. Mater Today 2025; 87: 403-421. [Article] [Google Scholar]
- Curry EJ, Ke K, Chorsi MT, et al. Biodegradable piezoelectric force sensor. Proc Natl Acad Sci USA 2018; 115: 909-914. [Article] [Google Scholar]
- Kalinin SV, Gruverman A. Scanning Probe Microscopy of Functional Materials: Nanoscale Imaging and Spectroscopy. New York: Springer, 2010. [Google Scholar]
- Ha PT, Lindemann SR, Shi L, et al. Syntrophic anaerobic photosynthesis via direct interspecies electron transfer. Nat Commun 2017; 8: 13924. [Article] [Google Scholar]
- Gao S, Lewis GD, Ashokkumar M, et al. Inactivation of microorganisms by low-frequency high-power ultrasound: 1. Effect of growth phase and capsule properties of the bacteria. Ultrasons SonoChem 2014; 21: 446-453. [Article] [Google Scholar]
- Pitt WG, Ross SA. Ultrasound increases the rate of bacterial cell growth. Biotechnol Prog 2003; 19: 1038-1044. [Article] [Google Scholar]
- Zhao B, Cui X, Ren W, et al. A controllable and integrated pump-enabled microfluidic chip and its application in droplets generating. Sci Rep 2017; 7: 11319. [Article] [Google Scholar]
- Stavenschi E, Corrigan MA, Johnson GP, et al. Physiological cyclic hydrostatic pressure induces osteogenic lineage commitment of human bone marrow stem cells: A systematic study. Stem Cell Res Ther 2018; 9: 276. [Article] [Google Scholar]
- Mulberry G, White K, Crocker M, et al. A 512-Ch dual-mode microchip for simultaneous measurements of electrophysiological and neurochemical activities. Biosensors 2023; 13: 502. [Article] [Google Scholar]
- Claassens NJ, Cotton CA, Kopljar D, Bar-Even A. Making quantitative sense of electromicrobial production. Nature Catalysis 2019; 2: 437-447. [Google Scholar]
- McCollom TM, Shock EL. Geochemical constraints on chemolithoautotrophic metabolism by microorganisms in seafloor hydrothermal systems. Geochim Cosmochim Acta 1997; 61: 4375-4391. [Google Scholar]
- An J, Kim B, Nam K, Lee Y, Chang I S. A pilot-scale benthic microbial electrochemical system (BMES) for enhanced organic removal in sediment restoration. Scient Report 2017; 7: 39802 [Google Scholar]
- Logan B E, Rabaey K. Conversion of wastes into bioelectricity and chemicals by using microbial electrochemical technologies. Science 2012; 337: 686-690. [CrossRef] [PubMed] [Google Scholar]
- Munk W, Wunsch C. Abyssal recipes II: Energetics of tidal and wind mixing. Deep-Sea Res Part I-Oceanogr Res Pap 1998; 45: 1977-2010. [Google Scholar]
- Stratford JP, Edwards CLA, Ghanshyam MJ, et al. Electrically induced bacterial membrane-potential dynamics correspond to cellular proliferation capacity. Proc Natl Acad Sci USA 2019; 116: 9552-9557. [Article] [Google Scholar]
- Devarajan A, Gupta D, Mitra K, et al. Chapter 11—Computational tools for design of synthetic genetic circuits. In: Singh V, ed. New Frontiers and Applications of Synthetic Biology. New York: Academic Press, 2022, 159–169. [Google Scholar]
- Andrianantoandro E, Basu S, Karig DK, et al. Synthetic biology: New engineering rules for an emerging discipline. Mol Syst Biol 2006; 2: 2006.0028. [Article] [Google Scholar]
- Yang Z, Zhou S, Zu J, et al. High-performance piezoelectric energy harvesters and their applications. Joule 2018; 2: 642-697. [Article] [CrossRef] [Google Scholar]
All Tables
All Figures
 |
Figure 1 Types of energy utilization by microorganisms. (A) Phototrophy; (B) chemotrophy; (C) electrotrophy; (D) piezo-electrotrophy. While all microorganisms involve electron transfer in metabolism and can be broadly considered electroactive, this article narrowly defines electrotrophic microorganisms as those directly acquiring electrons from external sources (e.g., minerals or electrodes) to emphasize piezoelectrotrophy’s uniqueness—generating/acquiring electrical energy from mechanical forces via piezoelectric mechanisms. |
| In the text | |
 |
Figure 2 Hypothetical energy flow in piezoelectrotrophy. This schematic depicts the proposed transduction pathway for piezoelectrotrophy, converting mechanical energy (left input) to bioavailable chemical energy (right output) via nano-bio interface processes. Mechanical stress induces shape changes in piezoelectric materials, causing charge separation and the generation of electrons (e−) and protons (H+) through the piezoelectric effect. Captured charges transfer across the microbial membrane via conductive nanowires, outer membrane cytochromes, or soluble mediators, establishing a membrane potential and proton gradient. The membrane potential and proton gradient drive ATP synthesis by ATP synthase or the reduction of electron carriers like NAD(P)H, integrating with cellular metabolism for growth and maintenance. The mechanism distinguishes piezoelectrotrophy from established paradigms by utilizing ubiquitous mechanical forces in energy-limited environments. |
| In the text | |
 |
Figure 3 Material types and metabolic categories in representative piezo-electrotrophy studies. The references related to piezoelectric materials in the image include struvite [37], BaTiO3 [1], PVDF [35], and diphenylalanine [36]. The references related to metabolic processes in the image include carbon fixation [1,38], sulfur Reduction [1], organic degradation [1], cell growth [1], metal reduction, and denitrification [1,37]. |
| In the text | |
 |
Figure 4 Ecological implications and technological applications. (A) Speculative presence in extreme environments; (B) potential role in biogeochemical cycles and ecosystem functions: driving forces behind Earth’s matter cycling; (C) significance in dark, oligotrophic environments: new insights into microbial survival strategies; (D) coexistence with other energy harvesting strategies (mixed trophic systems); (E) bioelectronic devices powered by piezoelectrotrophic systems; (F) environmental sensing and monitoring applications; (G) bioremediation possibilities using mechanical energy in contaminated sites; (H) energy harvesting from ambient mechanical sources (vibrations, fluid flow, pressure). |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.

