| Issue |
Natl Sci Open
Volume 5, Number 2, 2026
|
|
|---|---|---|
| Article Number | 20250058 | |
| Number of page(s) | 5 | |
| Section | Life Sciences and Medicine | |
| DOI | https://doi.org/10.1360/nso/20250058 | |
| Published online | 10 February 2026 | |
PERSPECTIVE
Cross-kingdom biological interactions regulate carbon source-sink in mangroves
1
Archaeal Biology Center, Synthetic Biology Research Center, Institute for Advanced Study, Shenzhen University, Shenzhen 518060, China
2
Shenzhen Key Laboratory of Marine Microbiome Engineering, Key Laboratory of Marine Microbiome Engineering of Guangdong Higher Education Institutes, Institute for Advanced Study, Shenzhen University, Shenzhen 518060, China
3
College of Life Sciences and Oceanography, Shenzhen University, Shenzhen 518060, China
* Corresponding author (email: This email address is being protected from spambots. You need JavaScript enabled to view it.
)
Received:
22
September
2025
Revised:
5
November
2025
Accepted:
3
February
2026
Mangrove wetlands, widely distributed in tropical and subtropical coastal areas, represent a critical component of “blue carbon” ecosystems due to their efficiency for carbon sequestration. The total global mangrove area is about 1.54×107 ha, with an estimated 3.7–6.2 Pg of carbon stock in 2020 [1]. Globally estimated carbon accumulation rate of sediments was about 48.52–59.01 Tg C yr−1 [2], while global mangrove CO2 emission was reported as 34.1 ± 5.4 Tg C yr−1 [3]. It is important to note that the estimation is highly uncertain. This perspective argues that this uncertainty stems from a critical gap: the failure to integrate the complex interactions spanning vegetation, benthic fauna, microorganisms (including both cellular Bacteria and Archaea and non-cellular viruses). Traditional research has focused on understanding individual biological groups in isolation. We propose a novel conceptual framework that the mangrove carbon source-sink is regulated by a cross-kingdom biological interaction network (Figure 1). We introduce key, testable hypotheses about how processes like the “Faunal Engineering” [4], “Microbial Carbon Pump” [5] and “Viral Shunt” [6] interact to control carbon fate. Deciphering these biological controls is the next frontier in blue carbon research, critical for refining predictive models of mangrove carbon budgets and advancing biotechnological strategies to amplify blue carbon potential.
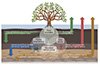 |
Figure 1 The cross-kingdom biological interactions driving carbon fate in mangrove ecosystems. Downward and upward arrows represent the pathways of carbon sequestration (e.g., carbon stabilization, burial and conversion into recalcitrant microbial residue) and emission (e.g., carbon decomposition and conversion into dissolved organic carbon), respectively. The ecosystem carbon balance is an outcome of these dynamic processes. The colors denote the different biological effects on carbon: green for plants, blue for fauna, brown for microorganisms, and red for viruses. |
Mangrove plants efficiently convert atmospheric CO2 into organic compounds, which are subsequently used to form plant biomass. Notably, mangrove leaf photosynthetic rates significantly exceed those of many tropical evergreen broad-leaved forests. This heightened photosynthetic efficiency enables rapid carbon assimilation, thereby enhancing their potential as a carbon sink [7]. Net ecosystem production (NEP) of mangroves ranges from 622.5 to 832.8 g C m−2 yr−1, which was regulated by a combination of global solar radiation, air temperature, and tidal inundation time [8]. Mangrove plants do more than fix CO2, and they create the niche for downstream organisms. Root exudates deliver labile organic carbon that primes the surrounding sediment and stimulates the decomposition of existing sediment organic matter. Root exudates can also lead to the stabilization of carbon as mineral-associated or microbial-derived organic matter [9]. Mangrove ecosystems comprise 84 plant species with 70 true mangrove species and 14 associated species [10]. We hypothesize that the carbon sequestration efficiency of mangroves is influenced by tree species composition, mediated by the distinct chemical profile of their root exudates and litter. Different species recruit specific microbial consortia, thereby directly influencing whether the rhizosphere functions as a net carbon source or sink.
Mangrove ecosystems are critical habitats for diverse fauna, including annelids, mollusks, crustaceans, mudskipper fish, birds such as kingfisher and heron, and even mammals like sea otters [11]. Keystone bioturbators, such as clams, polychaete worms and fiddler crabs, are not passive residents but actively mediate sediment biogeochemistry through multifaceted interactions [12]. Notably, sesarmid crabs transport 4.1 ± 2.2 Mg ha−1 yr−1 of fallen leaves into burrows, accounting for up to 87% of total mangrove litter turnover [13]. This activity efficiently moves carbon from the surface, where it is vulnerable to tidal export and rapid aerobic decomposition, into anoxic sediments conducive to long-term burial. Conversely, their burrowing introduces oxygen into the anoxic layer, and their excretion provides nutrients, stimulating microbial respiration and the oxidation of previously stable carbon. This influx of organic nutrients also stimulates methanogens in sub-oxide microsites and increases methane production by 80%–430% [14]. We propose that the net effect of bioturbation on carbon burial follows a particular relationship. There exists an optimal level of faunal activity that maximizes carbon sequestration by balancing litter burial with minimal sediment oxidation. Beyond this threshold, the mineralization pathway dominates, potentially flipping the system towards a net carbon source, especially when considering potent greenhouse gas (GHG) like methane.
The mangrove microbiome—comprising algae, fungi, bacteria, archaea, and viruses—mediates carbon flux through various mechanisms. Microorganisms are not just decomposers. They are also stabilizers. Through metabolic activity and cell death, they convert labile organic carbon into persistent, recalcitrant compounds (microbial residue carbon), which is known as Microbial Carbon Pump (MCP) [5]. Methane-oxidizing bacteria at the sediment-water interface oxidize methane, thereby preserving carbon storage in the Blue Carbon pool [15]. Viruses, particularly bacteriophages, which influence microbial mortality and metabolic pathways through lytic and lysogenic cycles, are a powerful disruptor [16]. Viral lysis of microbial hosts shunts carbon away from the microbial biomass and into the dissolved organic carbon (DOC) pool [17]. This “viral shunt” makes carbon readily available for heterotrophic respiration, potentially increasing GHG emissions. Furthermore, viral-encode auxiliary metabolic genes (AMGs), particularly carbohydrate-active enzymes (CAZymes), have been shown in other systems to enhance the degradation of complex polysaccharides such as cellulose, chitin, and xylan—abundant components of mangrove biomass, increase labile carbon availability for microbial metabolism and subsequent GHG production [18]. The ecological significance of AMGs in situ is an active area of research, and their net impact on carbon cycling in mangroves requires empirical validation. We hypothesize that the efficiency of the MCP is inversely related to viral-induced mortality. Under conditions of high viral lysis, the MCP is short-circuited, favoring mineralization. In contrast, periods of viral lysogeny or low infection rates may allow for greater carbon stabilization through microbial biomass buildup and residue formation.
To transition from concept to prediction, we propose a structured and interdisciplinary research agenda aimed at testing the hypothesis outlined in our perspective (Figure 2). First, to examine plant-sediment-microbe interactions, we will employ field mesocosms in which tree species composition is manipulated. This will be combined with 13C isotopic tracing of plant litter and DNA-based stable isotope probing (DNA-SIP) to identify active microbial decomposers and quantify carbon partitioning among CO2, microbial biomass and residues. Second, to assess the sediment-fauna-microbe interactions, a study manipulating crab presence/absence will be carried out. This will involve coordinated measurements of microbial community structure, faunal bioturbation intensity, and GHG flux. Third, to investigate phage-bacteria-climate interactions, we will conduct laboratory incubations of mangrove sediments under different temperature regimes. Using a metagenomic and metatranscriptomic approach, we will track viral replication strategies (lytic vs. lysogenic) and AMGs expression, alongside continuous GHG flux measurements. Data generated from these hypothesis-driven studies must be integrated into process-based biogeochemical models. Incorporating new modules that represent plant-microbe feedback, faunal bioturbation and viral lysis will substantially enhance the predictive accuracy of blue carbon models. This step is essential for translating mechanistic insights into actionable intelligence to support conservation and restoration.
 |
Figure 2 A framework for deciphering the biological controls of the mangrove carbon source-sink. This conceptual map bridges key hypotheses with a research agenda that combines experimental approaches (Mesocosm, Incubation, DNA-SIP) with advanced analytical techniques. The data generated will feed into process-based models, enabling robust forecasting of blue carbon stocks and fluxes. MCP: Microbial Carbon Pump; SIP: Stable Isotope Probing. |
Funding
This work was supported by the Guangdong Major Project of Basic and Applied Basic Research (2023B0303000017), the National Natural Science Foundation of China (32225003, 32393970, 32393971, 42476113, and 92251306), the Shenzhen Science and Technology Program (KCXFZ20240903092800002), the Project of Department of Education of Guangdong Province (2025KCXTD039), the Shenzhen University 2035 Program for Excellent Research (2022B002), the Scientific Foundation for Youth Scholars of Shenzhen University (868-000001032174), and the research fund from Synthetic Biology Research Center of Shenzhen University.
Conflict of interest
The authors declare no conflict of interest.
References
- Ouyang X, Lee SY. Improved estimates on global carbon stock and carbon pools in tidal wetlands. Nat Commun 2020; 11: 317. [Article] [Google Scholar]
- Wang F, Sanders CJ, Santos IR, et al. Global blue carbon accumulation in tidal wetlands increases with climate change. Natl Sci Rev 2021; 8: nwaa296. [Article] [Google Scholar]
- Rosentreter JA, Maher DT, Erler DV, et al. Seasonal and temporal CO2 dynamics in three tropical mangrove creeks—A revision of global mangrove CO2 emissions. Geochim Cosmochim Acta 2018; 222: 729-745. [Article] [Google Scholar]
- Wu D, Du E, Eisenhauer N, et al. Global engineering effects of soil invertebrates on ecosystem functions. Nature 2025; 640: 120-129. [Article] [Google Scholar]
- Jiao N, Luo T, Chen Q, et al. The microbial carbon pump and climate change. Nat Rev Microbiol 2024; 22: 408-419. [Article] [Google Scholar]
- Shiah FK, Lai CC, Chen TY, et al. Viral shunt in tropical oligotrophic ocean. Sci Adv 2022; 8: eabo2829. [Article] [Google Scholar]
- Zhang Z, Luo X, Friess DA, et al. Stronger increases but greater variability in global mangrove productivity compared to that of adjacent terrestrial forests. Nat Ecol Evol 2024; 8: 239-250. [Article] [Google Scholar]
- Gou R, Buchmann N, Chi J, et al. Temporal variations of carbon and water fluxes in a subtropical mangrove forest: Insights from a decade-long eddy covariance measurement. Agric For Meteor 2023; 343: 109764. [Article] [Google Scholar]
- Fang Q, Lu A, Hong H, et al. Mineral weathering is linked to microbial priming in the critical zone. Nat Commun 2023; 14: 345. [Article] [Google Scholar]
- Pham T, Yokoya N, Bui D, et al. Remote sensing approaches for monitoring mangrove species, structure, and biomass: Opportunities and challenges. Remote Sens 2019; 11: 230. [Article] [Google Scholar]
- Rahman, Lokollo FF, Manuputty GD, et al. A review on the biodiversity and conservation of mangrove ecosystems in Indonesia. Biodivers Conserv 2024; 33: 875-903. [Article] [Google Scholar]
- Kristensen E, Valdemarsen T, Moraes PC, et al. Pneumatophores and crab burrows increase CO2 and CH4 emission from sediments in two Brazilian fringe mangrove forests. Mar Ecol Prog Ser 2022; 698: 29-39. [Article] [Google Scholar]
- Zhang F, Liu X, Chen J, et al. Differences in organic carbon accumulation in mangrove soils due to foraging by herbivorous crabs. Mar Environ Res 2024; 194: 106332. [Article] [Google Scholar]
- Qin G, Lu Z, Gan S, et al. Fiddler crab bioturbation stimulates methane emissions in mangroves: Insights into microbial mechanisms. Soil Biol Biochem 2024; 194: 109445. [Article] [Google Scholar]
- Zhang X, Zhang C, Liu Y, et al. Non-negligible roles of archaea in coastal carbon biogeochemical cycling. Trends Microbiol 2023; 31: 586-600. [Article] [Google Scholar]
- Zhu Y, Li R, Yan S, et al. Habitat- and lifestyle-dependent structural and functional characteristics of viruses in mangrove wetlands of different functional zonings. Environ Res 2024; 252: 119070. [Article] [Google Scholar]
- Bonetti G, Trevathan-Tackett SM, Carnell PE, et al. The potential of viruses to influence the magnitude of greenhouse gas emissions in an inland wetland. Water Res 2021; 193: 116875. [Article] [Google Scholar]
- Jin M, Guo X, Zhang R, et al. Diversities and potential biogeochemical impacts of mangrove soil viruses. Microbiome 2019; 7: 58. [Article] [Google Scholar]
© The Author(s) 2026. Published by Science Press and EDP Sciences.
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
All Figures
 |
Figure 1 The cross-kingdom biological interactions driving carbon fate in mangrove ecosystems. Downward and upward arrows represent the pathways of carbon sequestration (e.g., carbon stabilization, burial and conversion into recalcitrant microbial residue) and emission (e.g., carbon decomposition and conversion into dissolved organic carbon), respectively. The ecosystem carbon balance is an outcome of these dynamic processes. The colors denote the different biological effects on carbon: green for plants, blue for fauna, brown for microorganisms, and red for viruses. |
| In the text | |
 |
Figure 2 A framework for deciphering the biological controls of the mangrove carbon source-sink. This conceptual map bridges key hypotheses with a research agenda that combines experimental approaches (Mesocosm, Incubation, DNA-SIP) with advanced analytical techniques. The data generated will feed into process-based models, enabling robust forecasting of blue carbon stocks and fluxes. MCP: Microbial Carbon Pump; SIP: Stable Isotope Probing. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.

