| Issue |
Natl Sci Open
Volume 5, Number 2, 2026
|
|
|---|---|---|
| Article Number | 20250067 | |
| Number of page(s) | 13 | |
| Section | Materials Science | |
| DOI | https://doi.org/10.1360/nso/20250067 | |
| Published online | 02 December 2025 | |
RESEARCH ARTICLE
In-situ polymerized siloxane cages for an interface-stable gel polymer electrolyte in lithium-metal batteries
1
College of Chemistry & Chemical Engineering, Central South University, Changsha 410083, China
2
State Key Laboratory of Powder Metallurgy, Central South University, Changsha 410083, China
3
Changsha Research Institute of Mining and Metallurgy Co., Ltd., Changsha 410012, China
* Corresponding authors (emails: This email address is being protected from spambots. You need JavaScript enabled to view it.
(Benhua Wang); This email address is being protected from spambots. You need JavaScript enabled to view it.
(Bowei Ju); This email address is being protected from spambots. You need JavaScript enabled to view it.
(Libao Chen))
Received:
17
October
2025
Revised:
26
November
2025
Accepted:
29
November
2025
Abstract
The rising demand for high energy density and safe rechargeable batteries calls for alternatives beyond conventional lithium-ion systems. Lithium metal batteries (LMBs) offer ultrahigh theoretical capacity but face critical challenges, including interfacial instability, dendritic lithium growth, and degradation of high voltage cathodes. Herein, we design a gel polymer electrolyte (HPP) via in-situ polymerization, integrating a fluorinated polymer network with highly cross-linked polyhedral oligomeric silsesquioxane (POSS) cages. The three-dimensional network enhances mechanical strength and promotes uniform Li+ transport, effectively suppressing dendrite formation. Simultaneously, fluorine-rich components extend the electrochemical stability window and enable a robust lithium fluoride (LiF) rich interphase, stabilizing the cathode interface. Benefiting from this synergistic design, Li||Li symmetric cells exhibit stable cycling for over 4500 h, while full cells retain 79% capacity after 500 cycles at 0.5 C and 87% after 200 cycles at 1 C. This work offers a rational strategy for electrolyte design to regulate interfacial chemistry and accelerate the development of high-performance LMBs.
Key words: polyhedral oligomeric silsesquioxane / gel polymer electrolyte / in-situ polymerization / lithium metal battery / stable interface
© The Author(s) 2025. Published by Science Press and EDP Sciences.
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
INTRODUCTION
The rapid expansion of renewable energy technologies and the electric vehicle industry is driving an urgent demand for next-generation rechargeable batteries with high energy density, enhanced safety, and long service life. Conventional lithium-ion batteries (LIBs), which typically employ graphite anodes, are now approaching their theoretical energy density limit (~350 Wh kg−1) [1]. Although silicon-carbon composite anodes offer a promising route to high capacity, their severe volume expansions (~400%) during cycling remain a major obstacle to practical application [2]. As a result, lithium metal batteries (LMBs) have attracted considerable attention as a next-generation energy storage technology due to the ultrahigh theoretical specific capacity of lithium metal anodes (~3860 mAh g−1) [3].
Despite their advantages, the commercialization of LMBs remains challenging due to persistent interfacial instability [4,5]. The highly reactive lithium metal surface readily forms an unstable interface with the electrolyte, often leading to uncontrolled dendritic lithium growth [6]. Such dendrites not only accelerate capacity fading but can also penetrate the separator, causing internal short circuits and severe safety risks [7]. Conventional carbonate-based electrolytes are poorly suited to address these issues [8], as they are flammable and ineffective in stabilizing electrode interfaces. Moreover, high-voltage cathode materials such as Ni-rich layered oxides (e.g., NCM811), which are essential for achieving high energy density, suffer from structural degradation and transition metal dissolution during cycling, further accelerating performance decay [9,10].
To overcome these limitations, the development of electrolytes capable of simultaneously stabilizing both the anode and cathode is essential. The key to stabilizing both the cathode and anode resides in the formation of a robust interface at each electrode. The cathode interface primarily functions to preserve the structural integrity of cathode particles [11], whereas the anode interface is mainly responsible for suppressing dendrite growth [12], a critical measure to ensure long-term cycling stability. Gel polymer electrolytes (GPEs) have emerged as a promising class of materials in this regard. Their solid or quasi-solid nature enhances safety, while their chemically tunable structures offer a versatile platform for engineering interfacial properties. Previous studies have shown that incorporating high-modulus nanomaterials, such as polyhedral oligomeric silsesquioxane (POSS) [13] or lithium silicates (LixSiOy) [14], can reinforce the electrolyte matrix and mechanically suppress dendrite growth. Meanwhile, the introduction of fluorine-containing components can expand the electrochemical stability window and promote the formation of robust, inorganic-rich cathode-electrolyte interphase (CEI), often dominated by lithium fluoride (LiF), which effectively protects cathode surfaces [15,16].
Herein, we report the design of a novel gel polymer electrolyte (denoted as HPP) prepared via an in-situ polymerization strategy that integrates a fluorinated polymer network with highly cross-linked, high-modulus POSS cages. This architecture simultaneously addresses interfacial challenges on both electrodes: the three-dimensional polymer network formed by POSS facilitates rapid lithium ion (Li+) transport and mechanically inhibits dendrite propagation, while the fluorinated components enhance oxidative stability and promote the formation of a stable, protective CEI on the cathode. Benefiting from this synergistic design, Li||Li symmetric cells exhibit remarkable cycling stability, sustaining Li+ stripping/plating for over 4500 h. When applied in full cells, the HPP electrolyte enables outstanding electrochemical performance, retaining 79% of capacity after 500 cycles at 0.5 C with an average Coulombic efficiency of 99.90%, and maintaining 87% of capacity after 200 cycles even at a high rate of 1 C. This work provides valuable insights into the rational design of polymer electrolytes for achieving interfacial stability and advancing the development of high-energy LMBs.
RESULTS AND DISCUSSION
Design strategy and structural characterization
To enhance the high-voltage stability of gel polymer electrolytes and their interfacial compatibility with both electrodes, nano-siloxane cage-like organo-inorganic composite species were introduced. The incorporation of fluorine-containing units provides excellent oxidative stability, while alkoxyl chains facilitate Li+ transport, synergistically improving electrochemical and physicochemical performance. Fluorinated species have demonstrated broad potential in polymer electrolytes [17], where rational molecular design can effectively regulate ion transport and stabilize interfaces. POSS, featuring a rigid Si-O cage and tunable organic periphery, offers a robust framework for hybrid electrolyte construction [17–19]. Here, thiol-functionalized POSS was cross-linked via a thiol-ene click reaction [20,21], enabling precise control over network architecture and mechanical integrity. A ternary mixture of ethylene carbonate (EC), ethyl methyl carbonate (EMC), and fluoroethylene carbonate (FEC) served as plasticizers, where EC and EMC promote fast Li+ conduction, while FEC enhances interfacial stability [22]. Lithium bis(trifluoromethanesulfonyl)imide (LiTFSI) and lithium difluoro(oxalato)borate (LiDFOB) were employed as lithium salts. Although LiTFSI provides excellent thermal and chemical stability, it can corrode current collectors under high voltages [23]. The introduction of LiDFOB mitigates this issue, as its decompositions form a passivation layer that suppresses LiTFSI-induced corrosion [24,25].
Density functional theory (DFT) calculations were performed to rationalize the component selection at the atomic level. The highest occupied molecular orbital (HOMO) and lowest unoccupied molecular orbital (LUMO) of the electrolyte precursors indicate their redox tendencies during cycling. In general, a lower LUMO energy level corresponds to stronger electron affinity (better reducibility), favoring the formation of a stable solid electrolyte interphase (SEI) on the anode [26,27]. While a lower HOMO energy level implies enhanced oxidation resistance, it also prevents electrolyte decomposition under high voltage. As shown in Figure 1a, LiDFOB and hexafluorobutyric anhydride (HFBA) exhibit lower LUMO levels, enabling the preferential formation of LiF-rich SEI layers. The LiF-dominated SEI exhibits favorable performance, with recent studies suggesting that this may be due to the formation of a LiH-LiF solid solution involving non-fluorinated electrolyte components [28]. Other precursors such as EC and FEC also display lower LUMO energy levels, providing sufficient anti-oxidative stability for high-voltage NCM811 cathodes. Notably, although POSS exhibits a relatively low LUMO value (−6.104 eV), its oxidative behavior in the polymer matrix is not determined solely by monomeric LUMO levels. The –SH groups of POSS are prone to oxidation in the monomeric state [29,30], but they are consumed during the thiol-ene crosslinking process, thereby eliminating oxidative sensitivity. Electrostatic potential (ESP) analysis revealed that regions with higher electron density (red zones) correspond to stronger Li+ affinity and lower binding energy, as seen in the carbonyl oxygens and the alkoxy chains of poly(ethylene glycol) diacrylate (PEGDA) (Figure 1b). Electron localization function (ELF) analysis further confirmed that PEGDA alkoxy chains provide abundant Li+ migration sites (Figure 1c). The introduction of POSS increases steric hindrance and enhances three-dimensionality [31,32], which synergistically promotes Li+ migration, resulting in higher ionic conductivity (0.10 mS cm−1).
 |
Figure 1 (a) HOMO and LUMO energy levels of polymer monomers, plasticizers, and lithium salts. (b) ESP of PEGDA, HFBA, FEC, EC, and EMC. (c) Electron localization function of POSS, HFBA, and PEGDA. (d) Schematic diagrams of gel polymerization for HP and HPP. (e) Schematic representation of the three-dimensional gel structure of HPP and its internal cross-linked network. (f) Photos of HP and HPP polymers before and after combustion. |
The polymeric network obtained via in-situ polymerization is shown in Figure 1d, and the molecular synthesis route of the HPP polymer is shown in Figure S1. The opaque white appearance of the HPP gel indicates microphase separation, which promotes efficient Li+ conduction. Figure 1e depicts the internal three-dimensional cross-linked network of the POSS-based GPE, where the interconnected channels allow solvated Li+ to migrate rapidly, achieving an ionic conductivity of 0.1 mS cm−1 superior to HP (0.05 mS cm−1, Figure S2). Benefiting from the organic-inorganic hybrid architecture of POSS, the HPP gel also exhibits relatively superior flame retardancy (Figure 1f and Figure S3). Upon combustion testing, HPP retained significant residue and unburned gel, suggesting the formation of a flame-retardant carbon layer that inhibited further burning, whereas HP displayed poorer flame resistance.
Fourier-transform infrared (FTIR) spectra confirm the chemical transformations before and after polymerization (Figure 2a and Figure S4). The S–H stretching vibration of POSS (~2550 cm−1) disappear after polymerization, indicating complete thiol conversion. The C=C stretching vibration (~1630 cm−1) of HFBA and PEGDA disappears completely in HPP but remains partially in HP (~1640 cm−1), indicating more complete consumption of double-bond in HPP due to the efficient thiol-ene click reaction. This leads to a denser and more uniform cross-linked network. Impedance measurements further confirm that although polymerization increases resistance, the HPP electrolyte exhibits lower impedance than HP both after polymerization and cycling, validating the superior ionic pathways and interfacial stability of the three-dimensional polymer network.
 |
Figure 2 (a) FTIR spectra of polymer precursors and resulting polymers. (b) Ionic conductivity of HPP and HP electrolytes from 30 to 80 °C, with fitted Arrhenius curves. Chronoamperometric curves of HP (c) and HPP (d) electrolytes, with insets showing impedance before and after polarization. (e) Tafel curves of symmetric cells assembled with HPP and HP electrolytes. (f) Coulombic efficiency measured by the Aurbach method. CCD of HP (g) and HPP (h) electrolytes. |
Li+ transport properties
The migration behavior of Li+ was analyzed from thermodynamic, kinetic, and electrochemical perspectives. Symmetric cells with blocking electrodes were used for electrochemical impedance spectroscopy (EIS) measurements at varying temperatures (Figure S6), and ionic conductivity was calculated and fitted (Figure 2b). At 30 °C, HPP exhibited an ionic conductivity of 0.121 mS cm−1, nearly double that of HP (0.061 mS cm−1), and consistently outperformed HP across all temperatures, demonstrating a positive correlation between conductivity and temperature. The temperature dependence of ionic conductivity followed the Arrhenius model, with activation energies (Ea) of 0.085 eV for HPP and 0.104 eV for HP. Thermodynamic analysis confirms that the HPP electrolyte exhibits superior Li+ conduction.
Kinetic properties were evaluated using the Li+ transference number (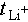 ) and exchange current density. A higher tLi+ reduces interfacial polarization, promotes uniform Li+ deposition, and improves cycling stability. As shown in Figure 2c and d, HPP exhibits a
) and exchange current density. A higher tLi+ reduces interfacial polarization, promotes uniform Li+ deposition, and improves cycling stability. As shown in Figure 2c and d, HPP exhibits a 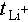 of 0.7, nearly double that of HP (0.45), primarily due to the continuous ion-transport channels in the HPP three-dimensional network. The exchange current density derived from Tafel plots further confirmed the kinetic advantage of HPP, with a value of 1.58 mA cm−2, almost four times that of HP (0.41 mA cm−2) (Figure 2e). Higher exchange current density may indicate a stable surface state and optimal thickness, improving lithium deposition and Coulombic efficiency. Aurbach-method measurements confirmed that HPP-based Li||Cu cells achieved an average Coulombic efficiency of 98.56%, compared to 95.88% for HP-based cells, a key factor for long-term cycling stability (Figure 2f). Critical current density (CCD) of Li||Li symmetric cells was tested to evaluate polarization voltage and dendrite suppression. As shown in Figure 2g and h, HP exhibited current fluctuations at 0.9 mA cm−2, whereas HPP maintained stable operation at 1.5 mA cm−2, demonstrating superior current tolerance and the ability to operate at higher current densities without short-circuiting.
of 0.7, nearly double that of HP (0.45), primarily due to the continuous ion-transport channels in the HPP three-dimensional network. The exchange current density derived from Tafel plots further confirmed the kinetic advantage of HPP, with a value of 1.58 mA cm−2, almost four times that of HP (0.41 mA cm−2) (Figure 2e). Higher exchange current density may indicate a stable surface state and optimal thickness, improving lithium deposition and Coulombic efficiency. Aurbach-method measurements confirmed that HPP-based Li||Cu cells achieved an average Coulombic efficiency of 98.56%, compared to 95.88% for HP-based cells, a key factor for long-term cycling stability (Figure 2f). Critical current density (CCD) of Li||Li symmetric cells was tested to evaluate polarization voltage and dendrite suppression. As shown in Figure 2g and h, HP exhibited current fluctuations at 0.9 mA cm−2, whereas HPP maintained stable operation at 1.5 mA cm−2, demonstrating superior current tolerance and the ability to operate at higher current densities without short-circuiting.
Li+ plating/stripping and lithium-metal interface stability
Next, we evaluated the electrochemical performance of the HPP electrolyte in practical applications through galvanostatic charge/discharge tests at varying current densities. Galvanostatic cycling at varying current densities revealed that HP exhibited a sharp increase in polarization current with increasing density, while HPP maintained a lower polarization voltage across the 0.025–0.6 mA cm−2 range, indicating superior Li+ plating/stripping behavior (Figure 3a). Long-term cycling tests on Li||Li symmetric cells further validated HPP’s superior cycling stability (Figure 3c and Figure S7). At a current density of 0.15 mA cm−2 and a capacity density of 0.15 mAh cm−2, HP achieved stable plating/stripping for only 1000 h (Figure 3b), while HPP exceeded 4500 h, maintaining a flat voltage platform. This enhanced stability is attributed to the lower thermodynamic energy and kinetic advantages of the three-dimensional polymeric network comprising high-modulus POSS.
 |
Figure 3 (a) Galvanostatic cycling of polymer electrolytes at different current densities. (b) Enlarged view of the short-circuit formation in HP electrolyte during long-term cycling of symmetric cells. (c) Continuous Li+ stripping/plating profiles of HPP and HP electrolytes at a current density of 0.15 mA cm−2. SEM images of the cycled lithium metal surface for HPP (d) and HP (e), along with corresponding EDS spectra of C, O, and F elements. XPS spectra of F 1s (f), C 1s (g), and Si 2p (h) for HPP and HP symmetric cells. |
To investigate the origins of long-term cycling stability and verify the polymer-lithium metal compatibility, we performed microscopic characterization. Scanning electron microscopy (SEM) was used to analyze the morphologies of cycled lithium metal surfaces and cross-sections, and energy-dispersive X-ray spectroscopy (EDS) provided elemental distributions. Figure 3d and e present the surface morphologies of HPP and HP, respectively. HPP exhibited a smooth, uniform morphology, while HP showed extensive uneven lithium dendrites. Even in the most severely damaged areas, dendrite growth in HPP was significantly suppressed compared to HP, confirming HPP effective dendrite suppression (Figure S9). EDS spectra also revealed a more homogeneous elemental distribution in HPP (Figure S10). Cross-sectional SEM analysis further corroborated HPP stable surface, where a uniform, flat SEI layer formed on its cross-section, in contrast to the disordered SEI layer in HP (Figure S11). Then, X-ray photoelectron spectroscopy (XPS) with depth profiling was performed to elucidate the SEI composition. The un-etched F 1s spectrum showed a dominant C–F bond (~688 eV) at the surface, which diminished after etching and was replaced by a LiF peak (~684.5 eV), indicating a robust LiF-rich SEI with an inorganic core and organic shell (Figure 3f). The weakened C–O bond signal further supported this (Figure S12). Although HP and HPP shared similar C and Li compositions, the key difference was in Si 2p spectra (Figure 3g and h). After etching, Si remained detectable in deeper layers of HPP (Si–O, ~103 eV), demonstrating that Si–O bonds in POSS contribute to the formation of a stable interface. The improved long-term cycling performance in symmetric batteries can be attributed to the formation of a stable LiF-dominated SEI on the lithium metal anode, which not only improves Li+ plating/stripping but also mitigates dendrite growth, thereby ensuring superior cyclic stability.
Performance of full-cell and interface compatibility
Before evaluating the long-term cycling performance of NCM811-based full cells, we first assessed the electrochemical window and leakage current of the electrolytes to verify their voltage compatibility with high-voltage cathodes. Linear sweep voltammetry (LSV) showed that HP had an electrochemical window of 4.73 V at 0.01 mA, while HPP exhibited a higher value of 5.25 V (Figure 4a). Both HP and HPP demonstrated wider electrochemical windows than conventional electrolytes, primarily due to the high-voltage tolerance of HFBA and FEC. The additional 0.52 V extension in HPP was attributed to the stable Si–O bonds in its POSS component, which facilitated the formation of a stable, high-voltage-tolerant three-dimensional cross-linked network. To further assess the voltage stability of the electrolytes, leakage current was evaluated via potentiostatic floating. At a voltage of 4.8 V, HP displayed a leakage current exceeding 20 μA (Figure 4b), whereas HPP remained stable with negligible leakage at 4.9 V, the upper detection limit of the instrument (Figure 4c).
 |
Figure 4 (a) LSV curves of HP and HPP electrolytes and their magnified views. Electropotential floating curves of HP (b) and HPP (c). (d) Cycling performance of Li||NCM811 cells with different polymer electrolytes at 0.5 C, along with corresponding Coulombic efficiency. Long-cycle capacity-voltage profiles of HP (e) and HPP (f) electrolytes from (d). (g) Rate capability of different polymer electrolytes. |
Having confirmed the high-voltage compatibility of the electrolytes, we next evaluated the electrochemical cycling performance of Li||NCM811 cells. As shown in Figure 4d, under room temperature, the Li||HPP||NCM811 cell cycled stably for 500 cycles at 0.5 C, retaining a capacity of 127.6 mAh g−1 (79% capacity retention), related cycling performance comparisons of Li||GPE||NCM are shown in Table S1, while the Li||HP||NCM811 cell retained only 71.1 mAh g−1(51% retention). HPP also exhibited a consistently high Coulombic efficiency, with an average of 99.90% (Figure S13), whereas HP showed more fluctuations and a lower Coulombic efficiency (99.82% average). These fluctuations in Coulombic efficiency indicate underlying interface instability, which ultimately impacts the battery cycle life. Figure 4e and f present the capacity-voltage profiles for HP and HPP at 0.5 C. After 500 cycles, HP exhibited significant capacity degradation, with its discharge voltage dropping to 3.9 V, indicating severe phase transitions in the HP cathode. Conversely, HPP maintained a higher capacity retention, with its discharge voltage only decreasing to 4.1 V. Rate capability testing revealed that HPP outperformed HP at all tested rates, achieving discharge capacities of 203.01, 194.85, 183.96, and 161.68 mAh g−1 at 0.05, 0.1, 0.2, and 0.5 C, respectively (Figure 4g). Notably, HPP exhibited superior reversibility when switching between high and low rates. These results emphasize the enhanced cycling stability and rate capability of HPP, confirming its potential for high-performance applications.
We then evaluated the long-term cycling performance of Li||NCM811 cells at 1 C rate to further confirm HPP advantages (Figure 5a). HPP exhibited an initial capacity of 136.59 mAh g−1 and retained 87% of its capacity after 200 cycles, while HP showed a lower initial capacity of 115.79 mAh g−1 and only 75% retention. Additionally, polymer compatibility with other cathodes was tested (Figures S14–S16), further demonstrating the potential of the HPP electrolyte for use in other cathode systems.
 |
Figure 5 (a) Cycling performance of Li||NCM811 cells using different electrolytes at 1 C and corresponding Coulombic efficiency. (b) SEM image of the cycled NCM811 cathode surface with different polymer electrolytes; inset shows a snapshot of the freshly disassembled cathode. (c) TEM images of surface particles of cycled NCM811 cathode with different polymer electrolytes. (d) TEM images of CEI on the surface of cycled NCM811 cathode particles with different polymer electrolytes. |
To explore the origin of this enhanced stability, SEM and transmission electron microscopy (TEM) were used to analyze the cathode surface. Figure 5b shows SEM images of cycled cathodes, with insets depicting post-cycling cathodes disassembled from the battery. Most HP cycled cathodes detached from the current collector and adhered to the separator, whereas HPP cycled cathodes remained intact on the current collector. SEM images also revealed more severe particles cracking in HP cathodes compared to HPP. Figure 5c presents detailed particle morphology. HPP secondary particles remained structurally intact and dense after cycling, while HP particles fragmented. The fragmentation of HP particles was likely due to unstable or non-uniform SEI formation on the cathode.
To further understand the differences in cycling performance, TEM analysis was performed on the CEI (Figure 5d). The results revealed that the CEI on HPP cycled cathodes was approximately 3 nm thick, with the cathode surface largely retaining its layered structure. In contrast, HP cycled cathodes exhibited a thicker (~8 nm) CEI, with more extensive rock-salt phase growth. This thicker, non-uniform CEI, combined with the rock-salt phase, likely hindered Li+ transport and contributed to the observed capacity fade [33]. These findings highlight the importance of stable CEI formation in enhancing long-term cycling stability, with HPP more uniform CEI playing a key role in its superior performance.
CONCLUSIONS
To summarize, we introduce siloxane cages with high modulus and high cross-linking properties into fluorinated polymer systems, creating a microphase-separated polymer electrolyte (HPP). The HPP electrolyte forms a robust three-dimensional cross-linked network that facilitates efficient Li+ migration while offering exceptional electrochemical stability, a high electrochemical window, and a low leakage current under high voltage. This electrolyte exhibits robust stability at both electrodes: on the cathode, it forms a uniform, dense, and long-lasting SEI dominated by LiF, enabling the Li||HPP||NCM811 cells to cycle stably for 500 cycles at a 0.5 C rate. On the anode, the high modulus of POSS suppresses dendrite formation, achieving over 4500 h of continuous lithium plating/stripping. Our design underscores the potential of high-modulus materials in polymer electrolytes, enabling enhanced electrode compatibility and addressing critical challenges in high-voltage battery performance.
MATERIALS AND METHODS
Materials
Lithium bis(trifluoromethanesulfonyl)imide (LiTFSI), lithium difluoro(oxalato)borate (LiDFOB), ethylene carbonate (EC), ethyl methyl carbonate (EMC), dimethyl carbonate (DMC), fluoroethylene carbonate (FEC), hexafluorobutyl acrylate (HFBA), azobisisobutyronitrile (AIBN, Aladdin), poly(ethylene glycol) diacrylate (PEGDA, Macklin), N-methyl-2-pyrrolidone (NMP, Aladdin), octamercaptopropyl polyhedral oligomeric- silsesquioxane (POSS-SH) (Aladdin), ethanol (Aladdin), polyvinylidene fluoride (PVDF), and conductive carbon (Super P) were purchased from Suzhou Duoduo Chemical Technology Co., Ltd., Aladdin, Macklin, and Guangdong Kangde New Energy Technology Co., Ltd., respectively. All chemicals were used without further purification. NCM811 and LFP cathode powders were obtained from Dongguan Kelude Innovation Technology Co., Ltd.
Cathode preparation: NCM811 cathodes were fabricated via a slurry coating method. The slurry, consisting of 80 wt% active material, 10 wt% Super P carbon (conductive agent), and 10 wt% PVDF (binder), was mixed in NMP for 24 h to form a homogeneous solution. The slurry was then coated onto aluminum foil, dried overnight in a forced-air oven (to prevent rapid NMP evaporation and bubble formation), and further dried in a vacuum oven at 80°C for 12 h, yielding cathodes with a mass loading of 2–3 mg cm−2. The same procedure was followed for LFP cathodes.
Characterization
FTIR spectroscopy was performed on a Thermo Scientific Nicolet 6700 instrument with a wavenumber range of 4000–400 cm−1. SEM and EDS were conducted on a JEOL JSM-7610FPlus to examine the morphology and elemental distribution of the lithium metal surface/cross-section. XPS (Thermo Scientific K-Alpha) with etching was used to analyze the composition and depth profile of the SEI on the lithium anode, with C–C bond reference peak fitting at 284.8 eV. TEM (JEOL JEM-F200) was used to investigate the cathode particle structure and the CEI. For TEM analysis, cathodes were extracted from full cells post-cycling, cleaned with DMC, lightly ground, ultrasonically dispersed in ethanol for 5 min, and deposited onto copper grids.
Ionic conductivity was measured by EIS using a Metrohm Autolab M204. Symmetric blocking cells with stainless steel electrodes were assembled, and tests were performed over 1–105 Hz with an AC amplitude of 10 mV. Ionic conductivity was calculated using Equation (1):
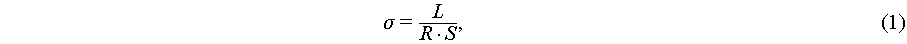 (1)
(1)
where L is the electrolyte thickness, R is the bulk resistance, and S is the contact area between the stainless steel and the polymer electrolyte.
For temperature-dependent conductivity, the same method was used, except with a frequency range of 0.1–106 Hz and 30 min pre-equilibration at each temperature. Ea was calculated via the Arrhenius Equation (2) [34]:
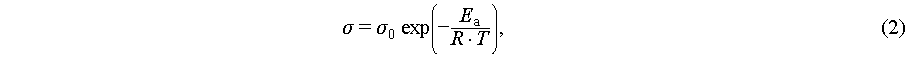 (2)
(2)
where σ0 is the pre-exponential factor, Ea is the activation energy (J mol−1 or kJ mol−1), T is the absolute temperature (K), and R is the molar gas constant (8.314 J mol−1 K−1).
The electrochemical window was determined using LSV at 1 mV s−1 (2–6 V). Tafel curves were obtained from a Li||Li symmetric cell on a CHI 660 workstation (1 mV s−1, −0.3–0.3 V) and fitted with CHI 660 software. 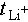 was measured via EIS and direct current polarization on a Li||Li symmetric cell (5 mV polarization voltage). The calculation used Equation (3):
was measured via EIS and direct current polarization on a Li||Li symmetric cell (5 mV polarization voltage). The calculation used Equation (3):
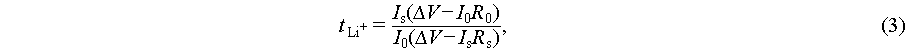 (3)
(3)
where Is is the steady-state current, I0 is the initial current, R0 is the interfacial resistance before polarization, and Rs is the interfacial resistance after polarization.
Coulombic efficiency was evaluated using a Li||Cu cell and the Aurbach method [35].
Data availability
The original data are available from corresponding authors upon reasonable request.
Funding
This work was supported by the National Natural Science Foundation of China (22578512, 52473204, U24A20501) and the Open Bidding for Selecting the Best Candidates in Department of Industry and Information Technology of Hunan Province (2024GXGG001).
Author contributions
H.C. conceived and designed the experiments and wrote the manuscript. C.F. analyzed the data. H.Z. and Y.W. provided the electrochemical-test protocols and reviewed the manuscript. Z.Z., Y.L. and D.W. participated in the experiments and in writing the manuscript. G.C.K. and B.W. initiated and designed the project. B.J., X.S. and L.C. provided funding and reviewed the manuscript.
Conflict of interest
The authors declare no conflict of interest.
Supplementary information
Supplementary file provided by the authors. Access Supplementary Material
The supporting information is available online at https://doi.org/10.1360/nso/20250067. The supporting materials are published as submitted, without typesetting or editing. The responsibility for scientific accuracy and content remains entirely with the authors.
References
- Xu C, Peng B, Yang W, et al. High energy density lithium battery systems: From key cathode materials to pouch cell design. Chem Soc Rev 2025; 54: 10245-10303. [Article] [Google Scholar]
- Chae S, Ko M, Kim K, et al. Confronting issues of the practical implementation of Si anode in high-energy lithium-ion batteries. Joule 2017; 1: 47-60. [Article] [Google Scholar]
- Xie Y, Huang Y, Zhang Y, et al. Surface modification using heptafluorobutyric acid to produce highly stable Li metal anodes. Nat Commun 2023; 14: 2883. [Article] [Google Scholar]
- Lin X, Xu S, Tong Y, et al. A self-healing polymerized-ionic-liquid-based polymer electrolyte enables a long lifespan and dendrite-free solid-state Li metal batteries at room temperature. Mater Horiz 2023; 10: 859-868. [Article] [Google Scholar]
- Liang W, Zhao K, Ouyang L, et al. A review of functional group selection and design strategies for gel polymer electrolytes for metal batteries. Mater Sci Eng-R-Rep 2025; 164: 100973. [Article] [Google Scholar]
- Meng YS, Srinivasan V, Xu K. Designing better electrolytes. Science 2022; 378: eabq3750. [Article] [Google Scholar]
- Niu H, Wang L, Guan P, et al. Recent advances in application of ionic liquids in electrolyte of lithium ion batteries. J Energy Storage 2021; 40: 102659. [Article] [Google Scholar]
- Guo JC, Chai CZ, Wang YH, et al. A fire-safe Li metal battery via smart gas management. Proc Natl Acad Sci USA 2025; 122: e2501549122. [Article] [Google Scholar]
- Zhang C, Zhao L, Li F, et al. Optimizing the electron density of PVDF-HFP-based solid polymer electrolyte by donor-acceptor COF toward high-performance solid-state lithium metal batteries. Adv Mater 2025; 37: e13427. [Article] [Google Scholar]
- Asl HY, Manthiram A. Reining in dissolved transition-metal ions. Science 2020; 369: 140-141. [Article] [Google Scholar]
- Kalnaus S, Dudney NJ, Westover AS, et al. Solid-state batteries: The critical role of mechanics. Science 2023; 381: eabg5998. [Article] [Google Scholar]
- Lv Q, Li L, Zhang X, et al. Developing dynamic ion transport channels in polymer solid electrolytes for high-performance lithium metal batteries. J Am Chem Soc 2025; 147: 27611-27623. [Article] [Google Scholar]
- Aranda K, Manthiram A. Influence of anode reactivity and chemical crossover on the formation of cathode-electrolyte interphase in high-nickel layered oxide cathodes. Adv Energy Mater 2025; : e02617. [Article] [Google Scholar]
- Huang Z, Lyu H, Greenburg LC, et al. Stabilizing lithium-metal electrodes with polymer coatings. Nat Energy 2025; 10: 811-823. [Article] [Google Scholar]
- Wang Y, Wu Z, Azad FM, et al. Fluorination in advanced battery design. Nat Rev Mater 2023; 9: 119-133. [Article] [Google Scholar]
- Wang Y, Li Z, Hou Y, et al. Emerging electrolytes with fluorinated solvents for rechargeable lithium-based batteries. Chem Soc Rev 2023; 52: 2713-2763. [Article] [Google Scholar]
- Tang L, Chen B, Zhang Z, et al. Polyfluorinated crosslinker-based solid polymer electrolytes for long-cycling 4.5 V lithium metal batteries. Nat Commun 2023; 14: 2301-2311. [Article] [Google Scholar]
- Cai T, Ma Z, Zhao F, et al. Interfacial chemistry of plasticizer to invoke high-performance silicon anodes for quasi-solid lithium-ion batteries. Adv Funct Mater 2025; : e15495. [Article] [Google Scholar]
- Kuo SW, Chang FC. POSS related polymer nanocomposites. Prog Polym Sci 2011; 36: 1649-1696. [Article] [Google Scholar]
- Dondoni A, Marra A. Recent applications of thiol-ene coupling as a click process for glycoconjugation. Chem Soc Rev 2012; 41: 573-586. [Article] [Google Scholar]
- Hu J, Wang W, Zhou B, et al. Click chemistry in lithium-metal batteries. Small 2024; 20: 2306622. [Article] [Google Scholar]
- Li M, Wang C, Davey K, et al. Recent progress in electrolyte design for advanced lithium metal batteries. SmartMat 2023; 4: e1185. [Article] [Google Scholar]
- Li Y, Ni Z, Geng J, et al. Advancements in electrolytes: From liquid to solid for low-cost and high-energy-density micro-sized silicon-based batteries. Adv Energy Mater 2025; 15: 2502284. [Article] [Google Scholar]
- Xu W, Dong W, Lin J, et al. Optimization design of fluoro-cyanogen copolymer electrolyte to achieve 4.7 V high-voltage solid lithium metal battery. Adv Sci 2024; 11: 2400466. [Article] [Google Scholar]
- Han C, Chen G, Ma Y, et al. Strategies towards inhibition of aluminum current collector corrosion in lithium batteries. Energy Mater 2023; 3: 300052. [Article] [Google Scholar]
- Adenusi H, Chass GA, Passerini S, et al. Lithium batteries and the solid electrolyte interphase (SEI)—Progress and outlook. Adv Energy Mater 2023; 13: 2203307. [Article] [Google Scholar]
- Wang A, Kadam S, Li H, et al. Review on modeling of the anode solid electrolyte interphase (SEI) for lithium-ion batteries. npj Comput Mater 2018; 4: 15. [Article] [Google Scholar]
- Liu X, Li S, Yuan C, et al. Probing the heterogeneous nature of LiF in solid-electrolyte interphases. Nature 2025; 646: 102-107. [Article] [Google Scholar]
- Willgert M, Kjell MH, Lindbergh G, et al. New structural lithium battery electrolytes using thiol-ene chemistry. Solid State Ion 2013; 236: 22-29. [Article] [Google Scholar]
- Li Z, Wang T, Zhong L, et al. Ultrathin thiol-ene crosslinked polymeric electrolyte for solid-state and high-performance lithium metal batteries. Sci China Mater 2023; 66: 1332-1340. [Article] [Google Scholar]
- Guan D, Yuan X, Li J, et al. Molecular-locking strategy enables volatile ether organic electrolytes to achieve high-energy lithium battery. Angew Chem Int Ed 2025; 64: e202517480. [Article] [Google Scholar]
- Lin J, Li Y, Wang Y, et al. POSS-crosslinked gel polymer electrolytes enabling low-temperature tolerant dye-sensitized solar cells. Electrochim Acta 2025; 513: 145596. [Article] [Google Scholar]
- Li Q, Liu G, Chen Y, et al. Electrolyte solvent-ion configuration deciphering lithium plating/stripping chemistry for high-performance lithium metal battery. Adv Funct Mater 2025; 35: 2420327. [Article] [Google Scholar]
- Dai C, Weng M, Cai B, et al. Ion-conductive crystals of poly(vinylidene fluoride) enable the fabrication of fast-charging solid-state lithium metal batteries. Energy Environ Sci 2024; 17: 8243-8253. [Article] [Google Scholar]
- Adams BD, Zheng J, Ren X, et al. Accurate determination of coulombic efficiency for lithium metal anodes and lithium metal batteries. Adv Energy Mater 2018; 8: 1702097. [Article] [Google Scholar]
All Figures
 |
Figure 1 (a) HOMO and LUMO energy levels of polymer monomers, plasticizers, and lithium salts. (b) ESP of PEGDA, HFBA, FEC, EC, and EMC. (c) Electron localization function of POSS, HFBA, and PEGDA. (d) Schematic diagrams of gel polymerization for HP and HPP. (e) Schematic representation of the three-dimensional gel structure of HPP and its internal cross-linked network. (f) Photos of HP and HPP polymers before and after combustion. |
| In the text | |
 |
Figure 2 (a) FTIR spectra of polymer precursors and resulting polymers. (b) Ionic conductivity of HPP and HP electrolytes from 30 to 80 °C, with fitted Arrhenius curves. Chronoamperometric curves of HP (c) and HPP (d) electrolytes, with insets showing impedance before and after polarization. (e) Tafel curves of symmetric cells assembled with HPP and HP electrolytes. (f) Coulombic efficiency measured by the Aurbach method. CCD of HP (g) and HPP (h) electrolytes. |
| In the text | |
 |
Figure 3 (a) Galvanostatic cycling of polymer electrolytes at different current densities. (b) Enlarged view of the short-circuit formation in HP electrolyte during long-term cycling of symmetric cells. (c) Continuous Li+ stripping/plating profiles of HPP and HP electrolytes at a current density of 0.15 mA cm−2. SEM images of the cycled lithium metal surface for HPP (d) and HP (e), along with corresponding EDS spectra of C, O, and F elements. XPS spectra of F 1s (f), C 1s (g), and Si 2p (h) for HPP and HP symmetric cells. |
| In the text | |
 |
Figure 4 (a) LSV curves of HP and HPP electrolytes and their magnified views. Electropotential floating curves of HP (b) and HPP (c). (d) Cycling performance of Li||NCM811 cells with different polymer electrolytes at 0.5 C, along with corresponding Coulombic efficiency. Long-cycle capacity-voltage profiles of HP (e) and HPP (f) electrolytes from (d). (g) Rate capability of different polymer electrolytes. |
| In the text | |
 |
Figure 5 (a) Cycling performance of Li||NCM811 cells using different electrolytes at 1 C and corresponding Coulombic efficiency. (b) SEM image of the cycled NCM811 cathode surface with different polymer electrolytes; inset shows a snapshot of the freshly disassembled cathode. (c) TEM images of surface particles of cycled NCM811 cathode with different polymer electrolytes. (d) TEM images of CEI on the surface of cycled NCM811 cathode particles with different polymer electrolytes. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.

