| Issue |
Natl Sci Open
Volume 5, Number 2, 2026
|
|
|---|---|---|
| Article Number | 20260005 | |
| Number of page(s) | 12 | |
| Section | Materials Science | |
| DOI | https://doi.org/10.1360/nso/20260005 | |
| Published online | 01 February 2026 | |
RESEARCH ARTICLE
Synthesis of one-dimensional hierarchical mesoporous carbon for supercapacitors
1
College of Energy Materials and Chemistry, Inner Mongolia Key Laboratory of Low Carbon Catalysis, College of Chemistry and Chemical Engineering, Inner Mongolia University, Hohhot 010070, China
2
Henan Institute of Advanced Technology, College of Chemistry, Zhengzhou University, Zhengzhou 450052, China
3
National Engineering Research Center for Marine Aquaculture, Institute of Innovation & Application, Zhejiang Ocean University, Zhoushan 316022, China
* Corresponding authors (emails: This email address is being protected from spambots. You need JavaScript enabled to view it.
(Danli Liang); This email address is being protected from spambots. You need JavaScript enabled to view it.
(Zaiwang Zhao); This email address is being protected from spambots. You need JavaScript enabled to view it.
(Yujuan Zhao))
Received:
8
January
2026
Revised:
29
January
2026
Accepted:
30
January
2026
Abstract
One-dimensional carbon-based materials suffer from inherent limitations such as structural collapse and high mass transfer resistance. In this study, a novel single-micelle-based dual-templates assembly strategy of using polystyrene-block-poly (4-vinylpyridine)-block-poly (ethylene oxide) (PS-PVP-PEO) single micelles as soft templates and one-dimensional (1D) SiO2 as hard templates was adopted. Utilizing dopamine as the carbon and nitrogen source, one-dimensional mesoporous nitrogen-doped carbon nanorods (1D N-mC) with hierarchical porous architecture were successfully prepared. Such hierarchical porous material features unique rich spherical pores with a specific surface area of 494 m2 g−1 and uniformly distributed nitrogen doping sites. Hence, this kind of 1D hierarchical mesoporous carbon material exhibits a specific capacitance of 320 F g−1 at 1 A g−1, a capacity retention rate of over 72% at a high rate of 10 A g−1, and only a 11% capacity decay after 5000 cycles. These properties effectively address the inherent defects aforementioned (structure collapse and high mass transfer resistance) of conventional carbon-based materials, laying a solid foundation for the development of high-performance energy storage devices.
Key words: one-dimensional / carbon-based materials / co-assembly / multi-level pore / supercapacitors
Contributed equally to this work.
© The Author(s) 2026. Published by Science Press and EDP Sciences.
 This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
This is an Open Access article distributed under the terms of the Creative Commons Attribution License (https://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
INTRODUCTION
In recent years, with the sustained growth of global energy demand and growing environmental issues, functional mesoporous materials with high-performance in energy storage and conversion have become a hot topic in the interdisciplinary fields [1–14]. This is because the mesoporous carbon materials not only possess mesoporous structural characteristics (rich pore channels) but also have the inherent advantages, such as good electrical conductivity [15–19]. Especially, the one-dimensional (1D) hierarchical porous carbon-based materials with their unique structural advantages have shown significant application potential in supercapacitors, catalysts, aerospace, automobiles, sports equipment, etc. [20–28]. Such hierarchical 1D mesoporous carbon nanoparticles with multi-level pore channels are potentially able to overcome the performance bottlenecks of traditional carbon materials and possess good capabilities in the regulation of properties [29–31].
In the past decade, some typical methods, involving emulsion-directed or hard templates, have been developed to synthesize the desirable one-dimensional hierarchical porous carbon-based materials, and some porous architectures have achieved [32–35]. For the emulsion-guided method, the obtained carbon materials lack pores and have an uneven pore structure. And for the conventional hard method, the pore structure is monotonous, and it is difficult to precisely control the pore channel structure (pore configuration) and construct multi-level pores. To date, for the synthesis of one-dimensional hierarchical porous carbon-based materials, traditional template methods have been continuously optimized while various new preparation approaches have emerged. Regarding performance modification, heteroatom doping and metal-based composite strategies have become the core modification approaches [36–39]; in terms of the application, they also performed good specific capacitance and rate performance in supercapacitors [40–42], demonstrating the application potential. However, traditional one-dimensional carbon materials are limited by their microporous structure and often suffer from high mass transfer resistance and uneven distribution of active sites, which restricts the improvement of their performance.
To address these issues, in this work, we propose a single-micelle-based dual-templates assembly strategy, combining the advantages of soft template (polystyrene-block-poly (4-vinylpyridine)-block-poly (ethylene oxide) (PS-PVP-PEO) micelles) and hard template (one-dimensional SiO2), to achieve the controllable preparation of one-dimensional hierarchical porous nitrogen-doped carbon materials. 1D N-mC is a hollow rod-shaped material with a length of ~350 nm. Its surface is decorated with spherical nanoparticles, exhibiting high roughness, and the inner surface contains micropores with a pore size of 1.0–2.5 nm. It features a three-level hierarchical interconnected structure of micropores, mesopores and macropores, with a specific surface area of 496 m2 g−1 and a pore volume of 0.62 cm3 g−1. The mesopore diameter is adjustable within 10–35 nm. Composed of C, O and N, it is rich in hydroxyl and other functional groups, exhibiting strong hydrophilicity. At a current density of 1 A g−1, its specific capacitance reaches 320 F g−1, retaining 270 F g−1 at 10 A g−1, with only 11% capacity decay after 5000 cycles.
RESULTS
Synthesis and characterization
One-dimensional hierarchical porous pore nitrogen-doped carbon materials were synthesized by the single-micelle-based dual-templates assembly strategy. The single micelle synthesized from PEO-PVP-PS was used as the template agent, and hydrochloric dopamine was used as the carbon source. Tetraethyl orthosilicate (TEOS) and cetyltrimethylammonium bromide (CTAB) were introduced (Figure 1a). First, one-dimensional SiO2 rods were prepared using CTAB as the structure-directing agent and TEOS as the silicon source. Then, the PS-PVP-PEO unimicellar ethanol solution was added dropwise to the SiO2 rod dispersion, and the micelles were assembled on the SiO2 surface at room temperature with stirring. Subsequently, the pH was adjusted, and dopamine hydrochloride was added. Afterwards, the mixture was stirred to enable dopamine nucleation and polymerization on the composite template. Next, the programmed calcination was performed in a nitrogen atmosphere to remove the micelles and carbonize the dopamine. Finally, SiO2 was etched with hydrofluoric acid to obtain the target one-dimensional hierarchical porous nitrogen-doped carbon nanorods (1D N-mC).
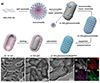 |
Figure 1 (a) Schematic illustration of the synthesis of one-dimensional hierarchical porous nitrogen-doped carbon materials; (b) SEM, (c, d) TEM, and (e) HAADF-STEM images, and (f–h) energy dispersive X-ray spectroscopy (EDX) elemental mapping images of the 1D N-mC material. |
Scanning electron microscopy (SEM) images show that it has a uniform rod-like structure with a length of approximately 350 nm, and a large number of spherical nanoparticles are distributed on the surface, with a relatively high surface roughness (Figure 1b); transmission electron microscopy (TEM) images further reveal that the material has a hollow nanorod structure, with a single-layer spherical mesopore on the surface without impurities, demonstrating excellent structural uniformity (Figure 1c). High-resolution TEM (HRTEM) shows that the diameter of the spherical mesopores is approximately 10 nm, with a wall thickness of 30 nm, and the mesopores are uniformly distributed over a wide range (Figure 1d); the medium-high angle annular dark-field (HAADF-STEM) images confirm that there are ultra-small pores of 1.0–2.5 nm on the inner surface of the material (Figure 1e), indicating that the ultra-small pores, large pores, and surface spherical mesopores are interconnected, forming a three-level system of “micro-pore-mesopore-large-pore” pore channels. The element mapping images show that the C, O, and N elements are uniformly distributed (Figure 1f, g, h), verifying the successful construction of a uniform nitrogen-doped carbon structure.
The nitrogen adsorption-desorption test indicates that the isotherm of 1D N-mC is a typical IV-type curve (Figure 2a). The adsorption amount sharply increases in the low-pressure region (P/P0 < 0.1), confirming the existence of micropores; the narrow-range hysteresis loop in the medium-pressure region (P/P0 ≈ 0.4–0.8) indicates uniform mesopore diameters, and the mesopore diameters calculated by the Barrett-Joyner-Halenda (BJH) model are concentrated at 12.3 nm (Figure 2b); the sudden increase in adsorption amount at the high-pressure region (P/P0 > 0.9) suggests the presence of macropores. The BET specific surface area of the porous material is high to 494 m2 g−1, and the total pore volume is 0.62 cm3 g−1, providing sufficient space for ion transport and exposure of active sites. The X-ray diffraction (XRD) spectrum shows a broadened (002) crystal plane diffraction peak at 25° (d ≈ 0.34 nm), indicating that the carbon material has many defects (Figure 2c); the Fourier transform infrared spectroscopy (FTIR) analysis shows that the broad peak at 3400 cm−1 corresponds to the N–H stretching vibration, the 1600 cm−1 peak belongs to the C=C skeleton vibration of the aromatic ring, and the multiple peaks in the 1000–1300 cm−1 range originate from the C–O–C/C–OH bond vibrations, confirming that the material surface is rich in hydroxyl, epoxy, and other oxygen-containing functional groups, and has strong hydrophilicity (Figure 2d).
 |
Figure 2 Characterization of mesoporous carbon materials. (a) N2 adsorption-desorption isotherm; (b) pore size distribution plot; (c) XRD pattern; (d) FTIR spectrum; (e–h) XPS spectra; (i) Raman spectrum. |
The X-ray photoelectron spectroscopy (XPS) full-spectrum scan confirms that 1D N-mC contains only three elements: C, O, and N, with high purity and no impurities (Figure 2e). Oxygen atom doping can alter the electron cloud distribution of carbon materials, enhance the electrical conductivity of the materials, and introduce active sites that are conducive to charge transfer (Figure 2f). The high-resolution C1s spectrum identifies characteristic peaks at 284.4 eV (C=C, sp2 carbon framework), 284.9 eV (C–N, nitrogen-doped carbon structure), and 290.7 eV (O–C=O, carboxylic acid group), indicating the presence of graphite-like carbon network, nitrogen-doped sites, and oxygen-containing functional groups in the material (Figure 2g); the N1s spectrum (Figure 2h) further distinguishes the forms of nitrogen, with coexistence of 398.1 eV (pyridine nitrogen), 400.2 eV (pyrrole nitrogen), 401.3 eV (graphite nitrogen), and 403.2 eV (oxidized nitrogen), providing abundant active sites and a controllable electronic structure for the material. The intensity ratio of the D band (1379 cm−1, disordered carbon/defect vibration) to the G band (1555 cm−1, sp2 carbon face internal stretching vibration) in the Raman spectrum is ID/IG = 1.06 (Figure 2i), confirming the presence of high-density defects and nanocrystalline boundaries in the carbon framework of the material, which is consistent with the XRD results.
The formation mechanism of 1D N-mC consists of three steps (Figure 3). The first step involves using micelles to form rod-shaped silica. CTAB acts as the cationic surfactant and self-assembles in a water solution to form rod-shaped micelles. TEOS hydrolyzes in an alkaline environment to generate silicic acid, which subsequently condenses to form SiO2 rods. After removing the CTAB micelles, mesoporous silica nanorods are obtained. The concentration of CTAB can control the morphology of SiO2. High concentration leads to an increase in diameter, while low concentration may result in the formation of spherical particles. The second step is the assembly of the single micelle on SiO2 interfaces. The single micelle of PS-PVP-PEO self-assembles into a core-shell-corona structure in ethanol (PS is the core, PVP is the shell, and PEO is the corona). The PVP segments of the PS-PVP-PEO micelles are positively charged and attract the negatively charged SiO2 surface through electrostatic attraction. The spatial steric hindrance effect of the PEO segments of micelles inhibits micelle aggregation, ensuring high monodispersity. The third step is dopamine coating and carbonization. The dopamine precursor polymerizes on the surface of the single micelle and SiO2 through electrostatic attraction due to spatial limitations. When the micelle and silica come into contact, dopamine cannot polymerize, forming pores. After carbonization and removal of the template, mesoporous structures were formed on the carbon shell, resulting in the one-dimensional mesoporous carbon nanorods.
 |
Figure 3 Formation mechanism of one-dimensional hierarchically porous carbon nanomaterials. |
Supercapacitor performance
To evaluate the influence of the aforementioned multi-level pore structure and nitrogen doping characteristics on the energy storage performance, a systematic electrochemical performance test of 1D N-mC was conducted for supercapacitors. Nano-carbon rods (N-CNR) and hollow nano-carbon rods (1D N-C) were employed as reference samples. Through a three-electrode system, cyclic voltammetry (CV) tests reveal that all samples exhibit typical double-layer capacitance characteristics, with CV curves approaching ideal rectangles (Figure 4a). Among them, the integral area of the CV curve of 1D N-mC is significantly larger than that of the reference samples, indicating the highest specific capacitance. When the scan rate is increased from 10 to 200 mV s−1, the CV curve of 1D N-mC is only slightly inclined without significant deformation (Figure 4b), demonstrating excellent rate performance [43,44]. In galvanostatic charge-discharge (GCD) tests, at a high current density of 10 A g−1, the charge-discharge curve of 1D N-mC remains symmetrical and isosceles triangles (Figure 4c), without significant voltage drop, further confirming its excellent charge storage capacity.
 |
Figure 4 Electrochemical performance of 1D N-mC in a three-electrode system. (a) Comparative CV curves of three materials at 10 mV s−1; (b) CV curves of 1D N-mC; (c) comparative GCD profiles of three materials at 2 A g−1; (d) GCD profiles of 1D N-mC; (e) amplified fit of impedance and equivalent circuit diagram; (f) specific capacitance comparison of three materials; (g) logarithmic current vs. logarithmic scan rate obtained in the cathodic direction; (h) typical charge storage contributions from capacitive and diffusion processes in 1D N-mC at 10 mV s−1 from cyclic voltammetry; (i) comparative histogram of percentage contributions from capacitive and diffusion-controlled charge storage in 1D N-mC at scan rates from 10 to 200 mV s−1. |
The electrochemical impedance spectroscopy (EIS) analysis reveals that the EIS Nyquist curve exhibits a small-diameter semicircle in the high-frequency region, corresponding to the small value of the low charge transfer resistance (Rct), indicating that 1D N-mC has excellent conductivity for rapid electron transfer throughout the electrode. The curve in the low-frequency region presents a steep and nearly vertical straight line, suggesting that the hierarchical pore structure of 1D N-mC can provide short and unobstructed diffusion channels for electrolyte ions. Additionally, the internal series resistance (Rs) within the 1D N-mC electrode is relatively low, which could also be demonstrated by the intercept at the real axis (Z′) of the middle EIS Nyquist curve (Figure 4e).
The specific capacitance (Figure 4f) was calculated based on the GCD curve. For 1D N-mC at current densities of 1, 2, 3, 4, and 5 A/g, the specific capacitance is 320, 310, 292.5, 280, and 272.5 F g−1 respectively, which is apparently higher than that of N-CNR and 1D N-C under the same conditions. Even at a high current density of 10 A/g, 1D N-mC still maintains a specific capacitance of 270 F g−1, with a capacity retention rate of 84.4%, while the capacity retention rates of 1D N-C and N-CNR are only 76.8% and 71.3%, respectively.
To further explore the charge storage mechanism, the reaction kinetics was analyzed through CV curves: According to the formula i = avb, the b value of 1D N-mC is 0.76–0.83 (Figure 4g), indicating that charge storage is mainly controlled by the capacitance process; further quantitative analysis through i = k1v + k2v1/2 shows that at a scan rate of 10 mV s−1, the contribution of diffusion control accounts for 76% of the total capacitance (Figure 4h), and with the scan rate increasing to 200 mV s−1, the contribution of surface capacitance increases to 59% (Figure 4i), confirming that the hierarchical pore structure can achieve efficient charge storage at different rates.
To further investigate the capacitive behavior of 1D N-mC, electrochemical tests were performed in a two-electrode configuration with 1 M Na2SO4 electrolyte, over a potential window of 0–1.3 V. When the voltage window increases from 0.5 to 1.2 V, the current density response shows a stepwise enhancement, but all CV curves display a rectangular shape with no significant changes. However, a slight peak is observed at 1.2–1.3 V, indicating that an oxygen reduction reaction occurred on the electrode. Therefore, the optimal voltage window for the symmetrical capacitor is determined to be 1.2 V (Figure 5a). Even at a high scan rate of 100 mV s−1, the CV curve still maintains a rectangular morphology (Figure 5b); the GCD test shows (Figure 5c) that the specific capacitance at 1 A g−1 was 205.8 F g−1, and it remains at 125 F g−1 at 10 A g−1, with a capacity retention rate of 60%. In the cycle stability test (Figure 5d), after cycling 5000 times at a current density of 10 A g−1, the initial capacitance is maintained at 89%, and the Coulomb efficiency was close to 100%.
 |
Figure 5 Electrochemical performance of 1D N-mC in symmetric supercapacitors. (a) CV curves at different voltage windows; (b) CV curves at various scan rates; (c) GCD profiles at different current densities; (d) cycling stability at 10 A g−1; (e) Ragone plot comparing this material with previously reported carbon-based supercapacitors in 1 M NaSO4 electrolyte. |
In the Ragone plot, the relationship between the energy density and power density of 1D N-mC symmetric capacitors is estimated [45–54]. It shows an energy density ranging from 41.2 to 25 Wh kg−1, and the weight power density increases from 600.5 to 6000 W kg−1, which is superior to most porous carbon-based symmetric supercapacitors previously reported (Figure 5e).
DISCUSSION
This study prepared one-dimensional hierarchical porous nitrogen-doped carbon materials (1D N-mC) through a single-micelle-based dual-templates assembly strategy. The correlation between the material structure and its properties was systematically explored, which was further addressed from the following two aspects.
(1) In terms of synthesis strategy, traditional hard/soft template methods for mesoporous carbon suffer from poor structural controllability and complex processes. This study innovatively combines the advantages of PS-PVP-PEO single micelle (soft template) and 1D SiO2 (hard template) via single-micelle-based dual-templates assembly strategy, precisely controlling mesopore size (10–40 nm) and balancing pore uniformity with structural stability. Uniform monolayered spherical mesopores are enabled by single micelles, and the 1D SiO2 hard template guides the fabrication of rod-like structures. Moreover, the subsequent etching process creates hollow channels to enhance mass transfer performance. Dopamine serves as a carbon/nitrogen source, realizing that the carbon skeleton construction and the nitrogen doping simultaneously provide abundant active sites. Controllable adjustment of mesopore size and SiO2 morphology is realized through the regulation of block copolymer molecular weight and CTAB concentration, furnishing a facile approach to the design of 1D hierarchical porous carbon.
For supercapacitor performance, 1D N-mC’s “micropore-mesopore-macropore” hierarchical structure exerts remarkable synergistic effects. For instance, micropores (494 m2 g−1 high specific surface area) provide abundant active sites; 12.3 nm mesopores can shorten the ion diffusion paths and reduce the mass transfer resistance; macropores and hollow structures facilitate electrolyte penetration and alleviate charge-discharge volume expansion. Additionally, nitrogen doping optimizes the performance: graphitic N enhances conductivity, pyridinic/pyrrolic N adds pseudocapacitance, and oxygen-containing groups boost hydrophilicity. Electrochemical tests show 84.4% capacity retention rate at 10 A g−1 and only 11% decay after 5000 cycles, outperforming N-CNR and 1D N-C.
CONCLUSIONS
This study focuses on the controllable preparation of one-dimensional hierarchical porous carbon materials for supercapacitors. A single-micelle-based dual-templates assembly strategy was proposed to address the structural defects and performance bottlenecks of traditional materials, successfully synthesizing one-dimensional hierarchical porous nitrogen-doped carbon materials (1D N-mC). The material has a high specific surface area of 494 m2 g−1. It possesses uniform mesopores (12.3 nm) and a three-level pore structure (micropore-mesopore-macropore). The mesopore size is tunable (10–35 nm) by adjusting the copolymer molecular weight. Nitrogen is uniformly distributed in the material in the forms of pyridinic nitrogen and graphitic nitrogen. As a supercapacitor electrode, 1D N-mC exhibits excellent performance: 320 F g−1 specific capacitance at 1 A g−1, 84.4% capacity retention at 10 A g−1, and only 11% decay after 5000 cycles. The symmetric supercapacitor based on this new porous architecture achieves high energy density (25–41.2 Wh kg−1) and power density (600.5–6000 W kg−1), outperforming most traditional carbon-based counterparts, which benefits from the synergistic effect of hierarchical pore-enhanced mass transfer and nitrogen doping-regulated electronics. This strategy provides a new approach for the structural design and performance optimization of carbon-based materials for energy storage.
Data availability
The original data are available from corresponding authors upon reasonable request.
Funding
This work was supported by the National Key Research and Development Program of China (2024YFE0101100) under its Singapore-China Joint Flagship Project (Clean Energy), the National Natural Science Foundation of China (22305132, 22475112 and 22365021), the Inner Mongolia Natural Science Foundation (2025LHMS02016), and the Open Research Fund of CNMGE Platform & NSCC-TJ (CNMGE2025001).
Author contributions
R.G. and B.L. conceived and designed the experiments and wrote the manuscript. X.F., D.L. and S.L. analyzed the data. X.C. and G.J. provided the electrochemical-test protocols and reviewed the manuscript. Y.D., P.Z., J.L., S.J.L. and S.S.L. participated in the experiments and in writing the manuscript. Y.Z. initiated and designed the project. Z.Z., Y.Z. and D.L. provided funding and reviewed the manuscript.
Conflict of interest
The authors declare no conflict of interest.
Supplementary information
Supplementary file provided by the authors. Access Supplementary Material
The supporting information is available online at https://doi.org/10.1360/nso/20260005. The supporting materials are published as submitted, without typesetting or editing. The responsibility for scientific accuracy and content remains entirely with the authors.
References
- Jiang M, Sun N, Li T, et al. Revealing the charge storage mechanism in porous carbon to achieve efficient K ion storage. Small 2024; 20: 2401478. [Article] [Google Scholar]
- Li J, Li R, Wang W, et al. Ordered mesoporous crystalline frameworks toward promising energy applications. Adv Mater 2024; 36: 2311460. [Article] [Google Scholar]
- Lee Y, Jo M, Kim J, et al. Versatile mesoporous microblocks prepared by pattern-induced cracking of colloidal films. Adv Mater 2023; 35: 2300952. [Article] [Google Scholar]
- Zhang R, Tang C, Yang S, et al. Double-walled mesoporous hydrogen-bonded organic frameworks with high methane storage capacity. J Am Chem Soc 2025; 147: 16412-16419. [Article] [Google Scholar]
- Chen S, Yuan L, Shi W. Structure-activity relationship of covalent organic frameworks (COFs) for gaseous iodine adsorption. Natl Sci Open 2024; : 20240026. [Article] [Google Scholar]
- Zhang X, Hao M, Yang X, et al. Promising porous materials for 99TcO4-removal from nuclear wastes. Natl Sci Open 2024; : 20240007. [Article] [Google Scholar]
- Zhao Y, Zhao Z. Monolayer mesoporous nanosheets with surface asymmetry via a dual-emulsion-directed monomicelle assembly. Chin J Struct Chem 2024; 43: 100238. [Article] [Google Scholar]
- Han X, Zhao Y, Zhao Z. A novel hydrogen-bonded mesoporous framework nanomaterials. Chem Synth 2024; 4: 81. [Article] [Google Scholar]
- Chen X, Zhao Z, Zhao Y. Uniform single-crystal mesoporous metal-organic frameworks with tunable architectures. Chin J Struct Chem 2025; 44: 100655. [Article] [Google Scholar]
- Duan L, Zhao Z. Construction of zeolite loaded ultra-small Ni nanoparticle catalysts. Chin J Struct Chem 2025; 44: 100516. [Article] [Google Scholar]
- Liu S, Zhao Y, Li S, et al. Design hydrophobic-internal and hydrophilic-external micropores for the preparation of microporous water. Chin J Struct Chem 2025; 44: 100701. [Article] [Google Scholar]
- He J, Sun Z, Huang L, et al. Two-dimensional conductive mesopore engineering of ultrahigh content covalent sulfur-doped carbon for superior sodium storage. J Mater Chem A 2025; 13: 15736-15742. [Article] [Google Scholar]
- Chen J, Li G, Bu F, et al. Tandem assembly and etching chemistry towards mesoporous conductive metal-organic frameworks for sodium storage over 50000 cycles. Angew Chem Int Ed 2025; 64: e202500287. [Article] [Google Scholar]
- Wang X, Zhang S, Li M, et al. Tailored design of mesoporous metal organic framework single crystals by kinetics-mediated micelle assembly for efficient asymmetrical single-atom catalysis. Adv Mater 2025; 37: 2500370. [Article] [Google Scholar]
- Zhang S, Li Y, Zhuang X, et al. Nano-metal-organic frameworks isolated in mesoporous structures. Adv Mater 2025; 37: 2418344. [Article] [Google Scholar]
- Luo W, Wang Y, Wang L, et al. Silicon/mesoporous carbon/crystalline TiO2 nanoparticles for highly stable lithium storage. ACS Nano 2016; 10: 10524-10532. [Article] [Google Scholar]
- Luo R, Zhang Z, Zhang J, et al. Bimetal CoNi active sites on mesoporous carbon nanosheets to kinetically boost lithium-sulfur batteries. Small 2021; 17: 2100414. [Article] [Google Scholar]
- Peng L, Hung CT, Wang S, et al. Versatile nanoemulsion assembly approach to synthesize functional mesoporous carbon nanospheres with tunable pore sizes and architectures. J Am Chem Soc 2019; 141: 7073-7080. [Article] [Google Scholar]
- Peng L, Peng H, Hung CT, et al. Programmable synthesis of radially gradient-structured mesoporous carbon nanospheres with tunable core-shell architectures. Chem 2021; 7: 1020-1032. [Article] [Google Scholar]
- Jiang H, Ma J, Li C. Mesoporous carbon incorporated metal oxide nanomaterials as supercapacitor electrodes. Adv Mater 2012; 24: 4196. [Article] [Google Scholar]
- Yang T, Wei L, Jing L, et al. Dumbbell-shaped Bi-component mesoporous janus solid nanoparticles for biphasic interface catalysis. Angew Chem Int Ed 2017; 56: 8459-8463. [Article] [Google Scholar]
- Hu K, Wang X, Hu Y, et al. Simultaneous improvement of oxygen reduction and catalyst anchoring via multiple dopants on mesoporous carbon frameworks for flexible Al-air batteries. ACS Nano 2022; 16: 19165-19173. [Article] [Google Scholar]
- Liu T, Yan M, Zhou S, et al. Site-selective superassembly of a multilevel asymmetric nanomotor with wavelength-modulated propulsion mechanisms. ACS Nano 2023; 17: 14871-14882. [Article] [Google Scholar]
- Xing Y, Xiu J, Zhou M, et al. Copper single-atom jellyfish-like nanomotors for enhanced tumor penetration and nanocatalytic therapy. ACS Nano 2023; 17: 6789-6799. [Article] [Google Scholar]
- Zhang H, Wang H, Pan Z, et al. Zn-metal-organic framework derived ordered mesoporous carbon-based nanostructure for high-performance and universal multivalent metal ion storage. Adv Mater 2022; 34: 2206277. [Article] [Google Scholar]
- Wang G, Cao Z, Gu D, et al. Nitrogen-doped ordered mesoporous carbon supported bimetallic PtCo nanoparticles for upgrading of biophenolics. Angew Chem Int Ed 2016; 55: 8850-8855. [Article] [Google Scholar]
- Saleem A, Chen J, Liu M, et al. Versatile magnetic mesoporous carbon derived nano-adsorbent for synchronized toxic metal removal and bacterial disinfection from water matrices. Small 2023; 19: 2207348. [Article] [Google Scholar]
- Trifonov A, Herkendell K, Tel-Vered R, et al. Enzyme-capped relay-functionalized mesoporous carbon nanoparticles: Effective bioelectrocatalytic matrices for sensing and biofuel cell applications. ACS Nano 2013; 7: 11358-11368. [Article] [Google Scholar]
- Zhang X, Lv X, Qian Z, et al. Template evolution induced relay self-assembly for mesoporous carbonaceous materials via hydrothermal carbonization. ACS Nano 2024; 18: 17826-17836. [Article] [Google Scholar]
- Li Y, Liu Y, Zhang K, et al. Ordered mesoporous carbon featuring single-crystal morphology and tunable pore architectures via EtOH-mediated micelle assembly. Small 2025; 21: 2502475. [Article] [Google Scholar]
- Zhou S, Bi W, Zhang J, et al. Strong interaction between titanium carbonitride embedded in mesoporous carbon nanofibers and Pt enables durable oxygen reduction. Adv Mater 2024; 36: 2400808. [Article] [Google Scholar]
- Wang C, Ma S, Wei Y, et al. Facile fabrication of monodisperse micron-sized dual Janus silica particles with asymmetric morphology and chemical environment. Small 2023; 19: 2208194. [Article] [Google Scholar]
- Lee SH, Lee B, Kim BJ, et al. Liquid precursor-assisted chemical vapor deposition of one-dimensional van der Waals material Nb2Se9: Tunable growth for room-temperature gas sensors. ACS Sens 2022; 7: 1912-1918. [Article] [Google Scholar]
- Luc W, Jiao F. Synthesis of nanoporous metals, oxides, carbides, and sulfides: Beyond nanocasting. Acc Chem Res 2016; 49: 1351-1358. [Article] [Google Scholar]
- Adler-Abramovich L, Marco P, Arnon ZA, et al. Controlling the physical dimensions of peptide nanotubes by supramolecular polymer coassembly. ACS Nano 2016; 10: 7436-7442. [Article] [Google Scholar]
- Zhao Z, Duan L, Zhao Y, et al. Constructing unique mesoporous carbon superstructures via monomicelle interface confined assembly. J Am Chem Soc 2022; 144: 11767-11777. [Article] [Google Scholar]
- Zhang J, Xia C, Su Y, et al. Boosted oxygen kinetics of hierarchically mesoporous Mo2C/C for high-current-density Zn-air battery. Small 2024; 20: 2307378. [Article] [Google Scholar]
- Zou Y, Zhou X, Zhu Y, et al. sp2-Hybridized carbon-containing block copolymer templated synthesis of mesoporous semiconducting metal oxides with excellent gas sensing property. Acc Chem Res 2019; 52: 714-725. [Article] [Google Scholar]
- Tian Z, Wang W, Dong C, et al. A general and scalable approach to sulfur-doped mono-/Bi-/trimetallic nanoparticles confined in mesoporous carbon. ACS Nano 2023; 17: 3889-3900. [Article] [Google Scholar]
- Pandolfo AG, Hollenkamp AF. Carbon properties and their role in supercapacitors. J Power Sources 2006; 157: 11-27. [Article] [Google Scholar]
- Feng LJ, Yan JY, Bian SW. Micro/mesoporous carbon nanospheres with homogeneously N,S,P-tridoped skeletons as electrodes for high-performance supercapacitors. ACS Appl Electron Mater 2023; 5: 6086-6093. [Article] [Google Scholar]
- Zhao C, Sun N, Li M, et al. Sn-improving flexible carbon electrode from electrospun nanofibers for a high-performance supercapacitor. Energy Fuels 2023; 37: 13467-13475. [Article] [Google Scholar]
- Zhi J, Zhao W, Liu X, et al. Highly conductive ordered mesoporous carbon based electrodes decorated by 3D graphene and 1D silver nanowire for flexible supercapacitor. Adv Funct Mater 2014; 24: 2013-2019. [Article] [Google Scholar]
- Chang CH, Chen KT, Hsieh YY, et al. Crystal facet and architecture engineering of metal oxide nanonetwork anodes for high-performance potassium ion batteries and hybrid capacitors. ACS Nano 2022; 16: 1486-1501. [Article] [Google Scholar]
- Qin X, Shan Z Q, Yuan Y, et al. A method for preparing carbon nanotubes or rods by hydrothermal synthesis. Chinese Patent. CN101254913A, 2008-09-03 [Google Scholar]
- Zhou H, Izumi J, Asano S, et al. Fast lithium intercalation mechanism on surface-modified cathodes for lithium-ion batteries. Adv Energy Mater 2023; 13: 2302402. [Article] [Google Scholar]
- Song J, Qi H, Yuan W, et al. Graphitic carbon nitride (g-C3N4) as an electrolyte additive boosts fast-charging and stable cycling of graphite anodes for Li-ion batteries. J Mater Chem A 2025; 13: 1964-1972. [Article] [Google Scholar]
- Zheng Z, Gao Q. Hierarchical porous carbons prepared by an easy one-step carbonization and activation of phenol-formaldehyde resins with high performance for supercapacitors. J Power Sources 2011; 196: 1615-1619. [Article] [Google Scholar]
- Xiong W, Liu M, Gan L, et al. A novel synthesis of mesoporous carbon microspheres for supercapacitor electrodes. J Power Sources 2011; 196: 10461-10464. [Article] [Google Scholar]
- Xu F, Tang Z, Huang S, et al. Facile synthesis of ultrahigh-surface-area hollow carbon nanospheres for enhanced adsorption and energy storage. Nat Commun 2015; 6: 7221. [Article] [Google Scholar]
- Wang J, Liu H, Zhang X, et al. Green synthesis of hierarchically porous carbon nanotubes as advanced materials for high-efficient energy storage. Small 2018; 14: 1703950. [Article] [Google Scholar]
- Wang JG, Liu H, Zhang X, et al. Elaborate construction of N/S-co-doped carbon nanobowls for ultrahigh-power supercapacitors. J Mater Chem A 2018; 6: 17653-17661. [Article] [Google Scholar]
- Huang Y, Zhou J, Gao N, et al. Synthesis of 3D reduced graphene oxide/unzipped carbon nanotubes/polyaniline composite for high-performance supercapacitors. Electrochim Acta 2018; 269: 649-656. [Article] [Google Scholar]
- Xing W, Qiao SZ, Ding RG, et al. Superior electric double layer capacitors using ordered mesoporous carbons. Carbon 2006; 44: 216-224. [Article] [Google Scholar]
All Figures
 |
Figure 1 (a) Schematic illustration of the synthesis of one-dimensional hierarchical porous nitrogen-doped carbon materials; (b) SEM, (c, d) TEM, and (e) HAADF-STEM images, and (f–h) energy dispersive X-ray spectroscopy (EDX) elemental mapping images of the 1D N-mC material. |
| In the text | |
 |
Figure 2 Characterization of mesoporous carbon materials. (a) N2 adsorption-desorption isotherm; (b) pore size distribution plot; (c) XRD pattern; (d) FTIR spectrum; (e–h) XPS spectra; (i) Raman spectrum. |
| In the text | |
 |
Figure 3 Formation mechanism of one-dimensional hierarchically porous carbon nanomaterials. |
| In the text | |
 |
Figure 4 Electrochemical performance of 1D N-mC in a three-electrode system. (a) Comparative CV curves of three materials at 10 mV s−1; (b) CV curves of 1D N-mC; (c) comparative GCD profiles of three materials at 2 A g−1; (d) GCD profiles of 1D N-mC; (e) amplified fit of impedance and equivalent circuit diagram; (f) specific capacitance comparison of three materials; (g) logarithmic current vs. logarithmic scan rate obtained in the cathodic direction; (h) typical charge storage contributions from capacitive and diffusion processes in 1D N-mC at 10 mV s−1 from cyclic voltammetry; (i) comparative histogram of percentage contributions from capacitive and diffusion-controlled charge storage in 1D N-mC at scan rates from 10 to 200 mV s−1. |
| In the text | |
 |
Figure 5 Electrochemical performance of 1D N-mC in symmetric supercapacitors. (a) CV curves at different voltage windows; (b) CV curves at various scan rates; (c) GCD profiles at different current densities; (d) cycling stability at 10 A g−1; (e) Ragone plot comparing this material with previously reported carbon-based supercapacitors in 1 M NaSO4 electrolyte. |
| In the text | |
Current usage metrics show cumulative count of Article Views (full-text article views including HTML views, PDF and ePub downloads, according to the available data) and Abstracts Views on Vision4Press platform.
Data correspond to usage on the plateform after 2015. The current usage metrics is available 48-96 hours after online publication and is updated daily on week days.
Initial download of the metrics may take a while.

